Chapter 1: Q. 1.56 (page 39)
Calculate the rate of heat conduction through a layer of still air that isthick, with an area of , for a temperature difference of .
Short Answer
The rate of heat conduction through a layer of still air is
Learning Materials
Features
Discover
Chapter 1: Q. 1.56 (page 39)
Calculate the rate of heat conduction through a layer of still air that isthick, with an area of , for a temperature difference of .
The rate of heat conduction through a layer of still air is
All the tools & learning materials you need for study success - in one app.
Get started for free
When spring finally arrives in the mountains, the snow pack may be two meters deep, composed of ice and of air. Direct sunlight provides about to earth's surface, but the snow might reflect of this energy. Estimate how many weeks the snow pack should last, if direct solar radiation is the only source of energy.
Consider a uniform rod of material whose temperature varies only along its length, in the direction. By considering the heat flowing from both directions into a small segment of length
derive the heat equation,
where , is the specific heat of the material, and is its density. (Assume that the only motion of energy is heat conduction within the rod; no energy enters or leaves along the sides.) Assuming that is independent of temperature, show that a solution of the heat equation is
where is a constant background temperature and is any constant. Sketch (or use a computer to plot) this solution as a function of , for several values of . Interpret this solution physically, and discuss in some detail how energy spreads through the rod as time passes.
The Fahrenheit temperature scale is defined so that ice melts at 320 F and water boils at 2120 F.
(a) Derive the formula for converting from Fahrenheit to Celsius and back
(b) What is absolute zero on the Fahrenheit scale?
An ideal diatomic gas, in a cylinder with a movable piston, undergoes the rectangular cyclic process shown in the given figure.
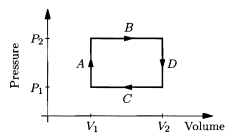
Assume that the temperature is always such that rotational degrees of freedom are active, but vibrational modes are "frozen out." Also assume that the only type of work done on the gas is quasistatic compression-expansion work.
(a) For each of the four steps A through D, compute the work done on the gas, the heat added to the gas, and the change in the energy content of the gas. Express all answers in terms of . (Hint: Compute before Q, using the ideal gas law and the equipartition theorem.)
(b) Describe in words what is physically being done during each of the four steps; for example, during step A, heat is added to the gas (from an external flame or something) while the piston is held fixed.
(c) Compute the net work done on the gas, the net heat added to the gas, and the net change in the energy of the gas during the entire cycle. Are the results as you expected? Explain briefly.
By applying Newton’s laws to the oscillations of a continuous medium, one can show that the speed of a sound wave is given by
,
where is the density of the medium (mass per unit volume) and B is the bulk modulus, a measure of the medium’s stiffness? More precisely, if we imagine applying an increase in pressure to a chunk of the material, and this increase results in a (negative) change in volume , then B is defined as the change in pressure divided by the magnitude of the fractional change in volume:
This definition is still ambiguous, however, because I haven't said whether the compression is to take place isothermally or adiabatically (or in some other way).
What do you think about this solution?
We value your feedback to improve our textbook solutions.