Chapter 6: Q 6.21 (page 233)
In the real world, most oscillators are not perfectly harmonic. For a quantum oscillator, this means that the spacing between energy levels is not exactly uniform. The vibrational levels of an H2 molecule, for example, are more accurately described by the approximate formula
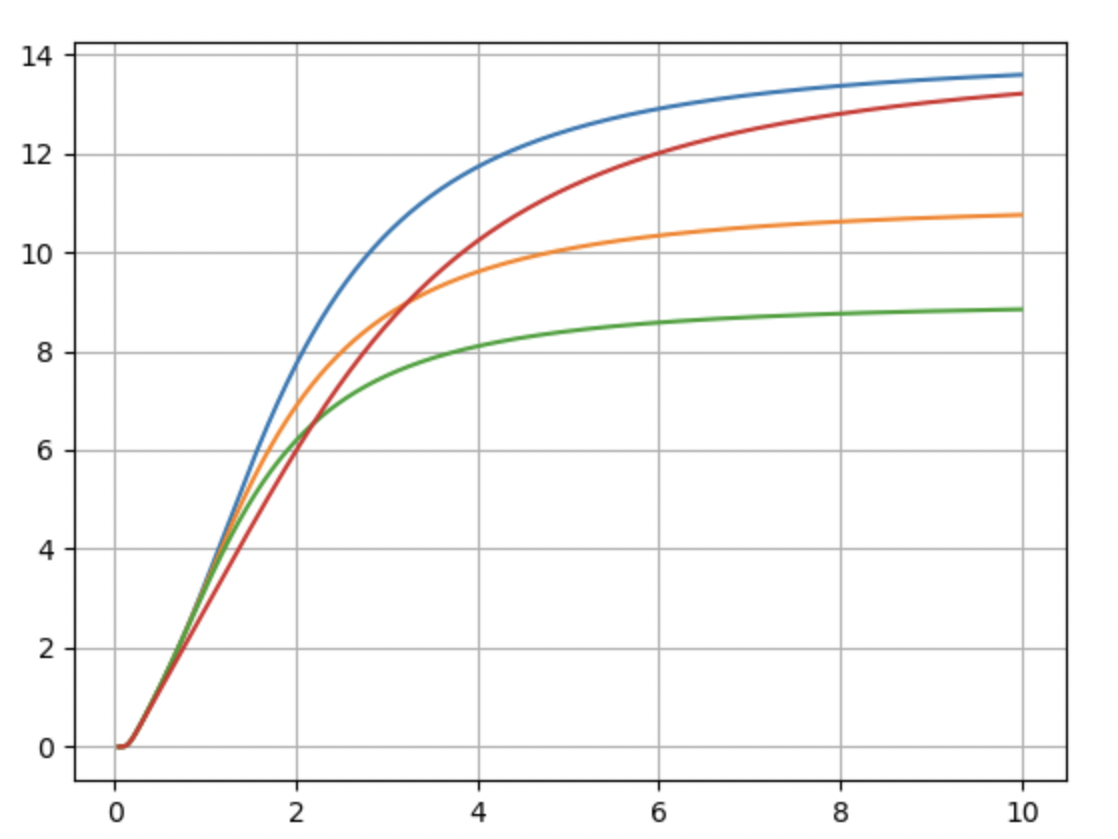
where is the spacing between the two lowest levels. Thus, the levels get closer together with increasing energy. (This formula is reasonably accurate only up to about n = 15; for slightly higher n it would say that En decreases with increasing n. In fact, the molecule dissociates and there are no more discrete levels beyond n 15.) Use a computer to calculate the partition function, average energy, and heat capacity of a system with this set of energy levels. Include all levels through n = 15, but check to see how the results change when you include fewer levels Plot the heat capacity as a function of . Compare to the case of a perfectly harmonic oscillator with evenly spaced levels, and also to the vibrational portion of the graph in Figure 1.13.
Short Answer
Therefore,