Chapter 10: Q. 35 (page 695)
In Problems 35–38, find parametric equations that define the curve shown.
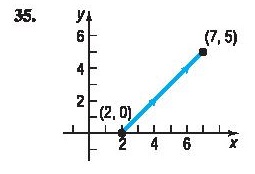
Short Answer
The parametric equation is
Learning Materials
Features
Discover
Chapter 10: Q. 35 (page 695)
In Problems 35–38, find parametric equations that define the curve shown.

The parametric equation is
All the tools & learning materials you need for study success - in one app.
Get started for free
Rutherford’s Experiment In May 1911, Ernest Rutherford published a paper in Philosophical Magazine. In this article, he described the motion of alpha particles as they are shot at a piece of gold foil 0.00004 cm thick. Before conducting this experiment, Rutherford expected that the alpha particles would shoot through the foil just as a bullet would shoot through snow. Instead, a small fraction of the alpha particles bounced off the foil. This led to the conclusion that the nucleus of an atom is dense, while the remainder of the atom is sparse. Only the density of the nucleus could cause the alpha particles to deviate from their path. The figure shows a diagram from Rutherford’s paper that indicates that the deflected alpha particles follow the path of one branch of a hyperbola.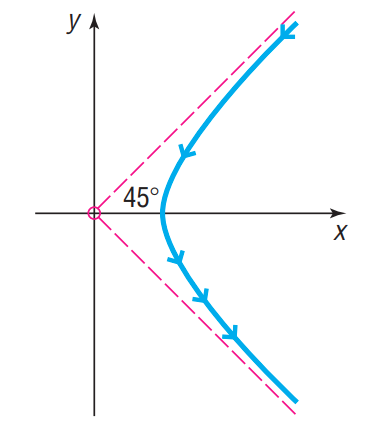
(a) Find an equation of the asymptotes under this scenario.
(b) If the vertex of the path of the alpha particles is cm from the center of the hyperbola, find a model that describes the path of the particle.
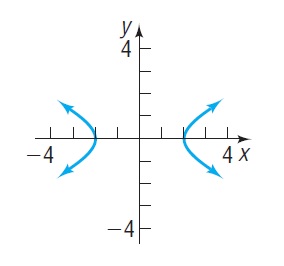
In Problems 15–18, the graph of a hyperbola is given. Match each graph to its equation.
Equations:
Graph:
The point that is symmetric with respect to the x-axis to the point (-2, 5) is ________.
True or False
To eliminate the term from the equation , rotate the axes through an angle , where .
Find the equation of the parabola described. Find the two points that define the latus rectum, and graph the equation by hand.
Focus at and vertex at.
What do you think about this solution?
We value your feedback to improve our textbook solutions.