Chapter 4: Q37P (page 93)
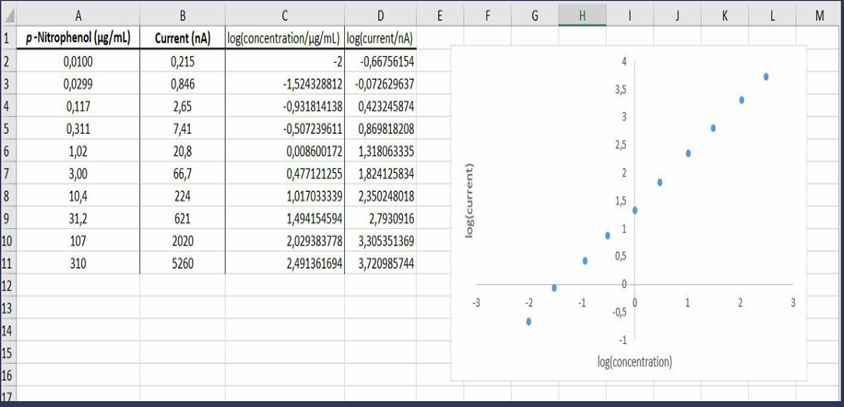
Logarithmic calibration curve. Calibration data spanning five orders of magnitude for an electrochemical determination of p-nitrophenol are given in the table. (The blank has already been subtracted from the measured current.) If you try to plot these dataon a linear graph extending from 0 to and from 0 to , most of the points will be bunched up near the origin. To handle data with such a large range, a logarithmic plot is helpful.
Overwhatrangeisthelog-logcalibrationlinear?
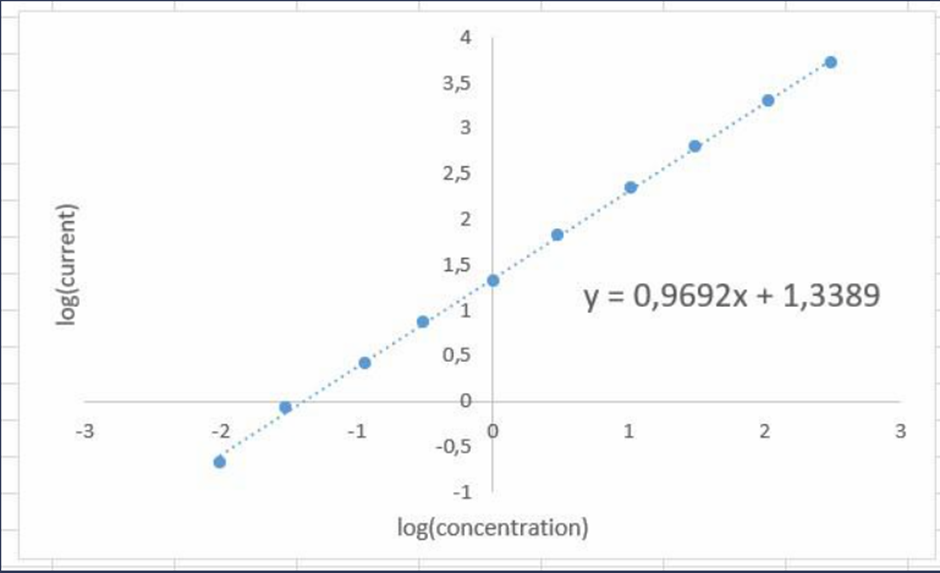
(a) Make a graph of log (current) versus log( concentration). Over what range is the log-log calibration linear?
(b)FindtheequationoftheLine
InTheform
(c) Find the concentration of p-nitrophenol corresponding to a signal of 99.9nA.
(d) Propagation of uncertainty with logarithm. For a signal of 99.9nA, log (concentration) and its standard uncertainty turn out to be . With rules for propagation of uncertainty from Chapter 3, find the uncertainty in concentration.
Short Answer
(a)The log-log calibration is linear all over.
(b)The equation of the line is:
(c)
(d) Propagation of uncertainty with logarithm is:
localid="1667561817319"