Chapter 4: Q24P (page 93)
Should the value 216 be rejected from the set of results 192,216,202,195, and 204?
Short Answer
The value 216 should not be rejected but has to be retained in the given results.
Learning Materials
Features
Discover
Chapter 4: Q24P (page 93)
Should the value 216 be rejected from the set of results 192,216,202,195, and 204?
The value 216 should not be rejected but has to be retained in the given results.
All the tools & learning materials you need for study success - in one app.
Get started for free
Traces of toxic, man-made hexachlorohexanes in North Sea sediments were extracted by a known process and by two new procedures, and measured by chromatography.
(a) Is the concentration parts per million, parts per billion, or something else?
(b) Is the standard deviation for procedure B significantly different from that of the conventional procedure?
(c) Is the mean concentration found by procedure B significantly different from that of the conventional procedure?
(d) Answer the same two questions as parts (b) and (c) to compare procedure A to the conventional procedure.
calibration curve based onknown points was used to measure the protein in an unknown. The results were protein , where the standard uncertainty is . Find the90%and 99%confidence intervals for protein in the unknown.
What is the relation between the standard deviation and the precision of a procedure? What is the relation between standard deviation and accuracy?
(a) The linear calibration curve in Figure 4-13 iswith. Find the quantity of unknown protein that gives a measured absorbance of when a blank has an absorbance of 0.095
(b) Figure 4-13 has n=14 calibration points in the linear portion. You measure k=14replicate samples of unknown and find a mean corrected absorbance of 0.169 Find the standard uncertainty and 95%confidence interval for protein in the unknown.
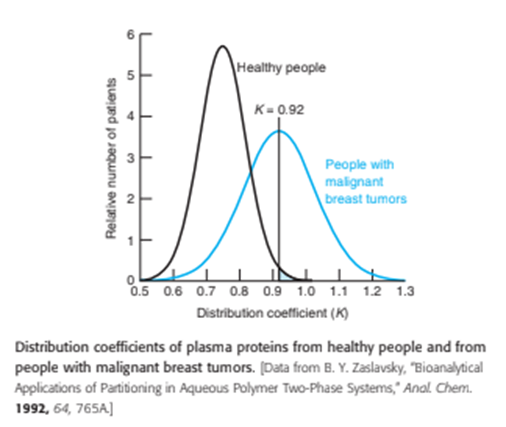
Blood plasma proteins of patients with malignant breast tumors differ from proteins of healthy people in their solubility in the presence of various polymers. When the polymers dextran and poly(ethylene glycol) are mixed with water, a two-phase mixture is formed. When plasma proteins of tumor patients are added, the distribution of proteins between the two phases is different from that of plasma proteins of a healthy person. The distribution coefficient ( K) for any substance is defined as K =[concentration of the substance in phase[concentration of the substance in phase B ]. Proteins of healthy people have a mean distribution coefficient of 0.75 with a standard deviation of 0.07. For the proteins of people with cancer, the mean is 0.92 with a standard deviation of 0.11.
(a) Suppose that Kwere used as a diagnostic tool and that a positive indication of cancer is taken as. What fraction of people with tumors would have a false negative indication of cancer because?
(b) What fraction of healthy people would have a false positive indication of cancer? This number is the fraction of healthy people with, shown by the shaded area in the graph below. Estimate an answer with Table 4 - 1 and obtain a more exact result with the NORMDIST function in Excel.
(c) Vary the first argument of the NORMDIST function to select a distribution coefficient that would identify 75% of people with tumors. That is, 75% of patients with tumors would have K above the selected distribution coefficient. With this value of K, what fraction of healthy people would have a false positive result indicating they have a tumor?
What do you think about this solution?
We value your feedback to improve our textbook solutions.