Chapter 28: Q3TY (page 775)
By what factor must the mass increase to reduce the sampling standard deviation by a factor of 2?
Short Answer
The mass needs to be increased four times to reduce the sampling standard deviation by a factor of 2.
Learning Materials
Features
Discover
Chapter 28: Q3TY (page 775)
By what factor must the mass increase to reduce the sampling standard deviation by a factor of 2?
The mass needs to be increased four times to reduce the sampling standard deviation by a factor of 2.
All the tools & learning materials you need for study success - in one app.
Get started for free
How does solid-supported liquid-liquid extraction differ from solid-phase extraction?
Barium titanate, a ceramic used in electronics, was analyzed by the following procedure: Into a Pt crucible was placed \(1.2\;{\rm{g}}\)of \({\rm{N}}{{\rm{a}}_2}{\rm{C}}{{\rm{O}}_3}\) and \(0.8\;{\rm{g}}\)of \({\rm{N}}{{\rm{a}}_2}\;{{\rm{B}}_4}{{\rm{O}}_7}\)plus \(0.3146\;{\rm{g}}\)of unknown. After fusion at \({1000^\circ }{\rm{C}}\)in a furnace for\(30\;{\rm{min}}\), the cooled solid was extracted with \(50\;{\rm{mL}}\)of\(6{\rm{MHCl}}\), transferred to a \(100 - {\rm{mL}}\) volumetric flask, and diluted to the mark. A \(25.00 - {\rm{mL}}\)aliquot was treated with \(5\;{\rm{mL}}\)of \(15\% \)tartaric acid (which complexes \({\rm{T}}{{\rm{i}}^{4 + }}\)and keeps it in aqueous solution) and \(25\;{\rm{mL}}\)of ammonia buffer,\({\rm{pH}}9.5\). The solution was treated with organic reagents that complex\({\rm{B}}{{\rm{a}}^{2 + }}\), and the \({\rm{Ba}}\)complex was extracted into \({\rm{CC}}{{\rm{l}}_4}.\)After acidification (to release the \({\rm{B}}{{\rm{a}}^{2 + }}\) from its organic complex), the \({\rm{B}}{{\rm{a}}^{2 + }}\)was backextracted into\(0.1{\rm{MHCl}}\). The final aqueous sample was treated with ammonia buffer and methylthymol blue (a metal ion indicator) and titrated with \(32.49\;{\rm{mL}}\) of \(0.01144{\rm{M}}\)EDTA. Find the weight per cent of Ba in the ceramic.
In 2002, workers at the Swedish National Food Administration discovered that heated, carbohydrate-rich foods, such as french fries, potato chips, and bread, contain alarming levels \((0.1to4\mu {\rm{g}}/{\rm{g}})\) of acrylamide, a known carcinogen\(36\).
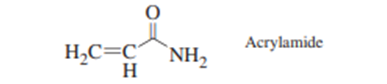
After the discovery, simplified methods were developed to measure ppm levels of acrylamide in food. In one procedure,\(10\;{\rm{g}}\) of pulverized, frozen french fries were mixed for \(20\;{\rm{min}}\)with\(50\;{\rm{mL}}\) of \({{\rm{H}}_2}{\rm{O}}\)to extract acrylamide, which is very soluble in water \((216\;{\rm{g}}/100\;{\rm{mL}}).\)The liquid was decanted and centrifuged to remove suspended matter. The internal standard \(^2{{\rm{H}}_3}\)-acrylamide was added to\(1\;{\rm{mL}}\) of extract. A solid-phase extraction column containing \(100{\rm{mg}}\)of cation-exchange polymer with protonated sulfonic acid groups\(\left( { - {\rm{S}}{{\rm{O}}_3}{\rm{H}}} \right.)\) was washed twice with 1 -mL portions of methanol and twice with \(1 - {\rm{mL}}\)portions of water. The aqueous food extract \((1{\rm{mL}})\)was then passed through the column to bind protonated acrylamide \(\left( { - {\rm{NH}}_3^ + } \right)\)to sulfonate \(\left( { - {\rm{SO}}_3^ - } \right)on\)the column. The column was dried for\(30\;{\rm{s }}at\)\(0.3\)bar and then acrylamide was eluted with\(1\;{\rm{mL}}\) of \({{\rm{H}}_2}{\rm{O}}.\)Eluate was analyzed by liquid chromatography with a polar bonded phase. The chromatograms show the results moni- tored by ultraviolet absorbance or by mass spectrometry. The retention time of acrylamide is different on the two columns because they have different dimensions and different flow rates.
(a) What is the purpose of solid-phase extraction prior to chromatography? How does the ion-exchange sorbent retain acrylamide?
(b) Why are there many peaks when chromatography is monitored by ultraviolet absorbance?
(c) Mass spectral detection used selected reaction monitoring (Figure 22-33) with the \(m/z72 \to 55\)transition for acrylamide and \(75 \to 58fo{r^2}{{\rm{H}}_3}\)-acrylamide. Explain how this detection method works and suggest structures for the ions with \({\rm{m}}/{\rm{z}}72\)and 55 from acrylamide.
(d) Why does mass spectral detection give just one major peak?
(e) How is the internal standard used for quantitation with mass spectral detection?
(f) Where does \(^2{{\rm{H}}_3}\)-acrylamide appear with ultraviolet absorbance? With mass spectral selected reaction monitoring?
(g) Why does the mass spectral method give quantitative results even though retention of acrylamide by the ion-exchange sorbent is not quantitative and elution of acrylamide from the sorbent by \(1\;{\rm{mL}}\) of water might not be quantitative?
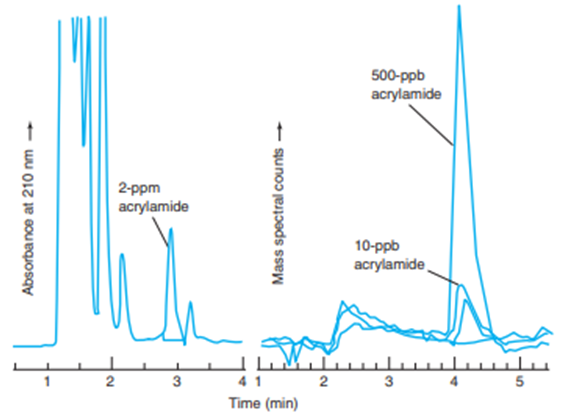
Chromatograms of acrylamide extract after passage through solid-phase extraction column. Left: Phenomenex Synergi Polar-RP 4- \(\mu {\rm{m}}\)column eluted with 96:4 \((vol/vol){{\rm{H}}_2}{\rm{O}}:{\rm{C}}{{\rm{H}}_3}{\rm{CN}}.\)Right: Phenomenex Synergi Hydro-RP 4- \(\mu {\rm{m}}\)column eluted with \(96:4:0.1(vol/vol/vol){{\rm{H}}_2}{\rm{O}}:{\rm{C}}{{\rm{H}}_3}{\rm{OH}}:{\rm{HC}}{{\rm{O}}_2}{\rm{H}}.\) (Data from L. Peng. T. Farkas, L. Loo, \({\rm{J}}.\)Teuscher, and \({\rm{K}}.\)Kallury, "Rapid and Reproducible Extraction of Acrylamide in French Fries Using a Single Solid-Phase Sorbent," Am. Lab. News Ed, October 2003, p. 10.)
Acid-base equilibria of Cr(III) were summarized in Problem 10-36. Cr(VI) in aqueous solution above pH 6 exists as the yellow tetrahedral chromate ion, \({\rm{CrO}}_4^{2 - }.\)Between\({\rm{pH}}2\)and \(6,{\rm{Cr}}\)(VI) exists as an equilibrium mixture of\({\rm{HCrO}}_4^ - \) and orange-red dichromate,\({\rm{C}}{{\rm{r}}_2}{\rm{O}}_7^{2 - }.{\rm{Cr}}({\rm{VI}})\) is a carcinogen, but \({\rm{Cr }}(III)\)is not considered to be as harmful. The following procedure was used to measure\({\rm{Cr }}({\rm{VI}})\) in airborne particulate matter in workplaces.
1. Particles were collected by drawing a known volume of air through a polyvinyl chloride filter with \(5 - \mu {\rm{M}}\)pore size.
2. The filter was placed in a centrifuge tube and \(10\;{\rm{mL}}\)of \(0.05{\rm{M}}{\left( {{\rm{N}}{{\rm{H}}_4}} \right)_2}{\rm{S}}{{\rm{O}}_4}/0.05{\rm{MN}}{{\rm{H}}_3}buffer,{\rm{pH}}8,\) were added. The immersed filter was agitated by ultrasonic vibration for\(30\;{\rm{min}}\)at \({35^\circ }{\rm{C}}\)to extract all \({\rm{Cr }}(III)and{\rm{Cr}}\)(VI) into solution.
3. A measured volume of extract was passed through a "strongly basic" anion exchanger (Table 26-1) in the \({\rm{C}}{{\rm{l}}^ - }\)form. Then the resin was washed with distilled water. Liquid containing \({\rm{Cr}}\)(III) from the extract and the wash was discarded.
4. Cr(VI) was then eluted from the column with\(0.5{\rm{M}}{\left( {{\rm{N}}{{\rm{H}}_4}} \right)_2}{\rm{S}}{{\rm{O}}_4}/0.05{\rm{MN}}{{\rm{H}}_3}\) buffer, \({\rm{pH}}8,\)and collected in a vial.
5. The eluted \({\rm{Cr}}\)(VI) solution was acidified with \({\rm{HCl}}\)and treated with a solution of 1,5 -diphenylcarbazide, a reagent that forms a colored complex with Cr(VI). The concentration of the complex was measured by its visible absorbance.
(a) What are the dominant species of \({\rm{Cr}}\)(VI) and \({\rm{Cr}}\)(III) at\({\rm{pH}}8\)?
(b) What is the purpose of the anion exchanger in step 3 ?
(c) Why is a "strongly basic" anion exchanger used instead of a "weakly basic" exchanger?
(d) Why is Cr(VI) eluted in step 4 but not step 3 ?
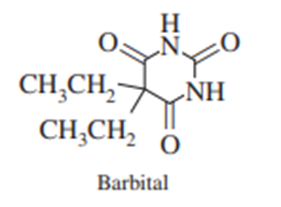
Barbital can be isolated from urine by solid-phase extraction with\({{\bf{C}}_{18}} - \)silica. The barbital is then eluted with\({\bf{1}}:{\bf{1}}\) vol/volacetone: chloroform. Explain how this procedure works.
What do you think about this solution?
We value your feedback to improve our textbook solutions.