Chapter 10: Q41P (page 232)
Explain how isoelectric focusing works.
Short Answer
Each of the proteins in the solution is focused in one region at their isoelectric
Learning Materials
Features
Discover
Chapter 10: Q41P (page 232)
Explain how isoelectric focusing works.
Each of the proteins in the solution is focused in one region at their isoelectric
All the tools & learning materials you need for study success - in one app.
Get started for free
Heterogeneous equilibrium.dissolves in water to give "carbonic acid" (which is mostly dissolved, as described in Box 6-4).
(The equilibrium constant is called the Henry's law constant for carbon dioxide, because Henry's law states that the solubility of a gas in a liquid is proportional to the pressure of the gas.) The acid dissociation constants listed for "carbonic acid" in Appendix G apply to. Given thatin the atmosphere is atm, find the of water in equilibrium with the atmosphere.
A solution was prepared from 10.0 mLof 0.100Mcacodylic acid and 10.0mLof 0.0800MNaOH. To this mixture was added 1.00mL of morphine. Calling morphine B, calculate the fraction of morphine present in the form .
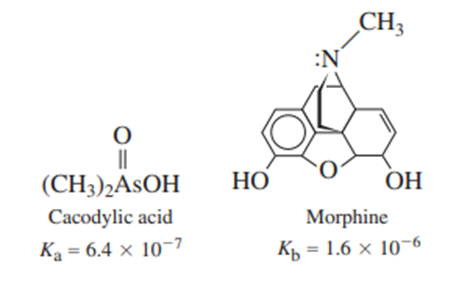
Consider , the intermediate form of a diprotic acid.for this species isandis. Nonetheless, the and reactions proceed to nearly the same extent when NaHA is dissolved in water. Explain.
(a) Calculate the quotient in .
(b) Find the same quotient for .
The both are equal as found to be
What do you think about this solution?
We value your feedback to improve our textbook solutions.