Chapter 22: Q42P (page 603)
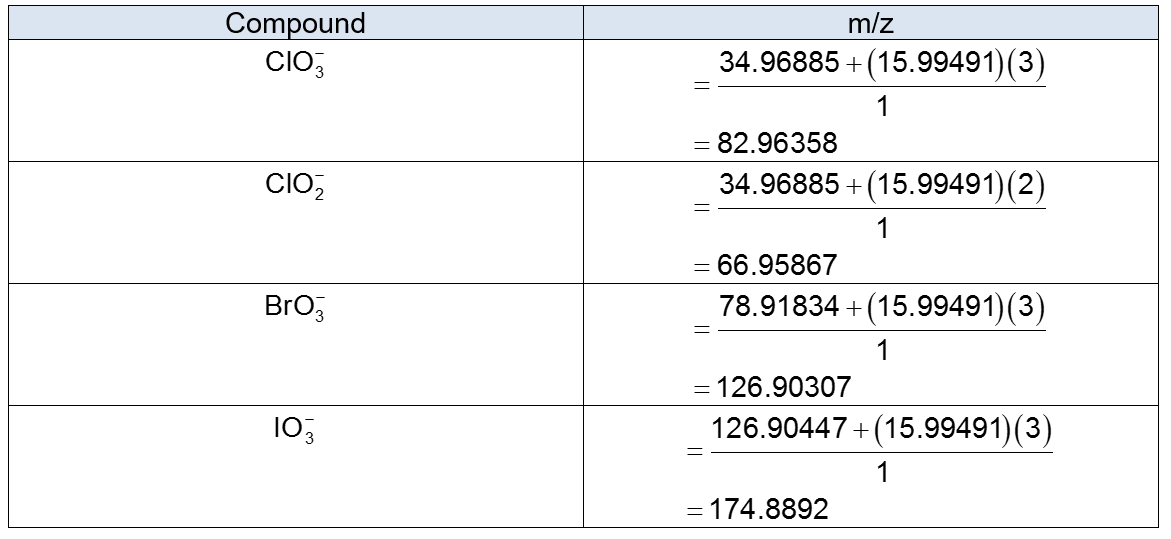
Chlorate , chlorite , bromate , and iodate can be measured in drinking water at the 1-ppb level with 1% precision by selected reaction monitoring. Chlorate and chlorite arise from used as a disinfectant. Bromate and iodate can be formed from or when water is disinfected with ozone . For the highly selective measurement of chlorate, the negative ion selected by Q1 in Figure 22-33 is m/z 83 and the negative ion selected by Q3 is m/z 67. Explain how this measurement works and how it distinguishes from , , and
Short Answer
The answer is not given in the drive.