Chapter 22: QCE (page 598)
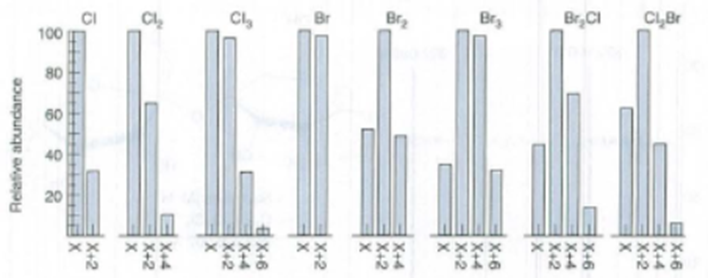
Consider an element with two isotopes whose natural abundances are aand . If there are n atoms of the element in a compound, the probability of finding each combination of isotopes is derived from the expansion of the binomial . For carbon, the abundances are are andfor . The probability of finding atoms in acetylene, HC = CH, is given by the first term of the expansion of . The value ofis, so the probability of finding $ atoms in acetylene is 0.9787. The probability of finding is . The probability of finding role="math" localid="1663661299432" is . The molecular ion, by definition, contains atoms. The M + 1 peak contains . The intensity of M + 1 relative to will be . (We are ignoring because its natural abundance is small.) in 1 ,2-dichlorobenzene. Draw a stick diagram of the distribution, like Figure 22 - 7.
Short Answer
The stick diagram: