Chapter 7: Q3TY (page 150)
Find 49.1 mL.
Short Answer
Thevalue for the titration of is 12.86.
Learning Materials
Features
Discover
Chapter 7: Q3TY (page 150)
Find 49.1 mL.
Thevalue for the titration of is 12.86.
All the tools & learning materials you need for study success - in one app.
Get started for free
Construct a graph of versus milliliters of for the titration of of solution containing and. The titrant is. Calculate at the following volumes:
localid="1654845944495" second equivalence point,.
Derive an expression analogous to Equation 7-12 for the titration of (concentration, ,volume=) with (titrant concentration=). Your equation should allow you to compute the volume of titrant as a function of.
Why does the surface charge of a precipitate change sign atthe equivalence point?
Ascorbic acid (vitamin C) reacts with according to the equation
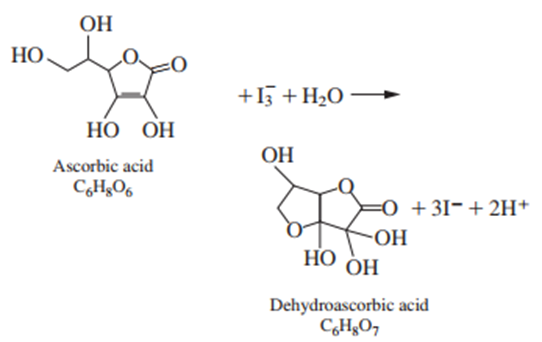
Starch is used as an indicator in the reaction. The end point is marked by the appearance of a deep blue starch-iodine complex when the first fraction of a drop of unreacted remains in the solution.
(a) Verify that the structures above have the chemical formulas written beneath them. You must be able to locate every atom in the formula. Use atomic masses from the periodic table on the inside cover of this book to find the formula mass of ascorbic acid.
(b) If 29.41 mL of solution are required to react with 0.197 0 g of pure ascorbic acid, what is the molarity of the solution?
(c) A vitamin C tablet containing ascorbic acid plus inert binder was ground to a powder, and 0.424 2 g was titrated by 31.63 mL of . Find the weight percent of ascorbic acid in the tablet.
A 25.00 - mL solution containing 0.031 10 M was titrated with 0.025 70 M to precipitate calcium oxalate: Find pCa2+ at the following volumes
of :
(a) 10.00,
(b) Ve
(c) 35.00 mL
What do you think about this solution?
We value your feedback to improve our textbook solutions.