Chapter 7: Q31P (page 160)
Why does the surface charge of a precipitate change sign atthe equivalence point?
Short Answer
There is a shift in sign beyond the equivalence point due to extra silver ions on the precipitate.
Learning Materials
Features
Discover
Chapter 7: Q31P (page 160)
Why does the surface charge of a precipitate change sign atthe equivalence point?
There is a shift in sign beyond the equivalence point due to extra silver ions on the precipitate.
All the tools & learning materials you need for study success - in one app.
Get started for free
Limestone consists mainly of the mineral calcite, .The carbonate content of 0.5413 g
powdered limestone was measured by suspending the powder in water, adding 10.00 mL of and heating to dissolve the solid and expel role="math" localid="1654937947195" Calcium carbonate FM 100.087
The excess acid required of for complete titration to a phenolphthalein end point. Find the weight percent of calcite in the limestone.
How does a blank titration reduce titration error?
Use Equation 7-12 to reproduce the curves in Figure 7-3. Plot your results on a single graph.
The text claims that precipitation of is not complete before begins to precipitate in the titration in Figure 7-4. Calculate the concentrationof at the equivalence point in the titration of alone. Show that this concentration of will precipitate.
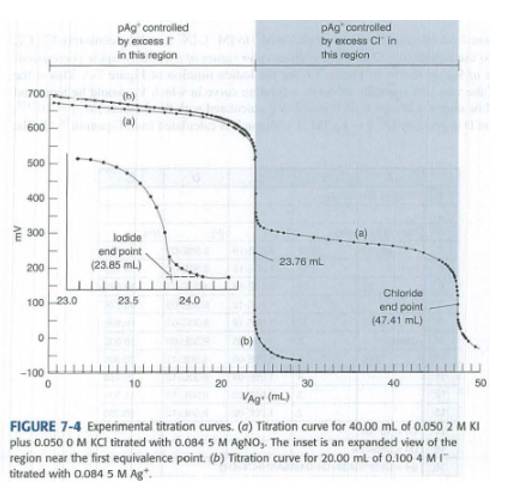
Distinguish between the terms equivalence point and end point.
What do you think about this solution?
We value your feedback to improve our textbook solutions.