Chapter 7: Q29P (page 160)
Consider precipitation of with role="math" localid="1655098271650" :
Write mass balance equations for M and X and derive the equation
Short Answer
The mass balance for M and X are.
Learning Materials
Features
Discover
Chapter 7: Q29P (page 160)
Consider precipitation of with role="math" localid="1655098271650" :
Write mass balance equations for M and X and derive the equation
The mass balance for M and X are.
All the tools & learning materials you need for study success - in one app.
Get started for free
What is the difference between a reagent-grade chemical and a primary standard?
: Describe the chemistry that occurs in each of the following
regions in curve (a) in Figure 7-4: (i) before the first equivalence
point; (ii) at the first equivalence point; (iii) between the first and
second equivalence points; (iv) at the second equivalence point; and
(v) past the second equivalence point. For each region except (ii),
write the equation that you would use to calculate .
Use Equation 7-12 to reproduce the curves in Figure 7-3. Plot your results on a single graph.
A solid mixture weighing 0.05485 gcontained only ferrous ammonium sulfate and ferrous chloride. The sample was dissolved in , and the required 13.39mLof for complete oxidation to. Calculate the weight percent CI ofin the original sample.
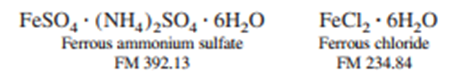
Consider the titration of 25.00 mL of 0.08230 M KI with 0.05110 M AgNO3. Calculate pAg+ at the following volumes of added AgNO3:
(a) 39.00 mL
(b) Ve
(c) 44.30 mL
What do you think about this solution?
We value your feedback to improve our textbook solutions.