Managing a salt-water aquarium. A tank at the New Jersey State Aquarihas
a volume of 2.9million liters.are used to remove nitrate
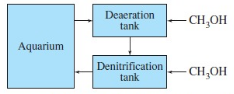
that would otherwise build up to toxic levels. Aquarium water is first pumped into
a 2700-Ldesecration tank containing bacteria that consumein the presence
of added methanol:
Anoxic (deoxygenated) water from the desecration tank flows into a gentrification reactor containing colonies of Pseudomonas bacteria in a porous medium. Methanol is injected continuously and nitrate is converted into nitrite and then into nitrogen:
a) Desecration can be thought of as a slow, bacteria-mediated titration
ofbylocalid="1655109035857" . The concentration oflocalid="1655109054860" in seawater at localid="1655109040090" is localid="1655109045111" .
How many liters oflocalid="1655109050648" are required
by Reaction 1for 2.9 million liters of aquarium water?
b) Write the net reaction showing nitrate plus methanol going to nitrogen.
How many liters of localid="1655109058452" are required by the net reaction for 2.9 million
liters of aquarium water with a nitrate concentration of localid="1655109062338" ?
c) In addition to consuming methanol for Reactions 1 through 3, the
bacteria require 30% more methanol for their own growth. What is the
total volume of methanol required to denitrify 2.9 million liters of
aquarium water?



