Chapter 23: QCE (page 628)
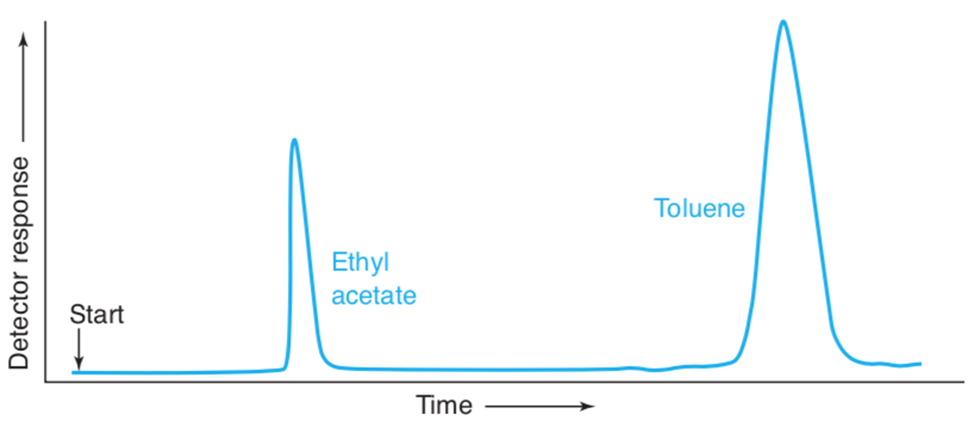
Find the retention factors for octane and nonane in figure 23-7. What you measure distances , estimate them to the nearest 0.1mm.
(b) Find the ratio
(c) Find the Relative retention for octane and nonane.
(d) Find the partition coefficient for Octane by assuming that the volume of the stationary phase equals half the volume of the mobile phase.
Short Answer
(a) Retention factor for octane, = 3.83
Retention factor for octane, = 7.31
(b)Ratio,
(c)Relative retention ratio ,
(d)Partition coefficient,