Chapter 23: QBE (page 628)
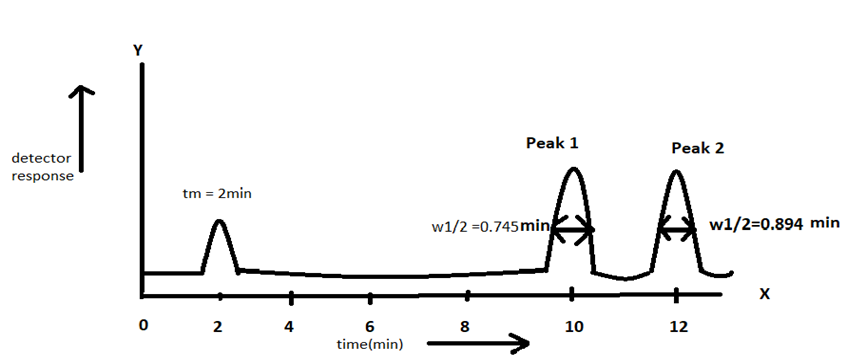
Consider a chromatography experiment in which two components with retention factors and are injected into column with theoretical plates. The retention time for the less-retained component is .
(a)Calculate and.Find (width at half height) and w (at the base) for each peak.
(b)Using graph paper , sketch the chromatogram analogous to figure 23-7,supposing that two peaks have same amplitude(height). Draw the half widths accurately.
(c)Calculate the resolution of the two peaks and compare this value with those drawn in Figure 23-10.
Short Answer
(a)
are 1.265 min and 1.518 min.
(for peak1 ) and (for peak2) are 0.745 min and 0.894 min
(b) Resolution =1.437