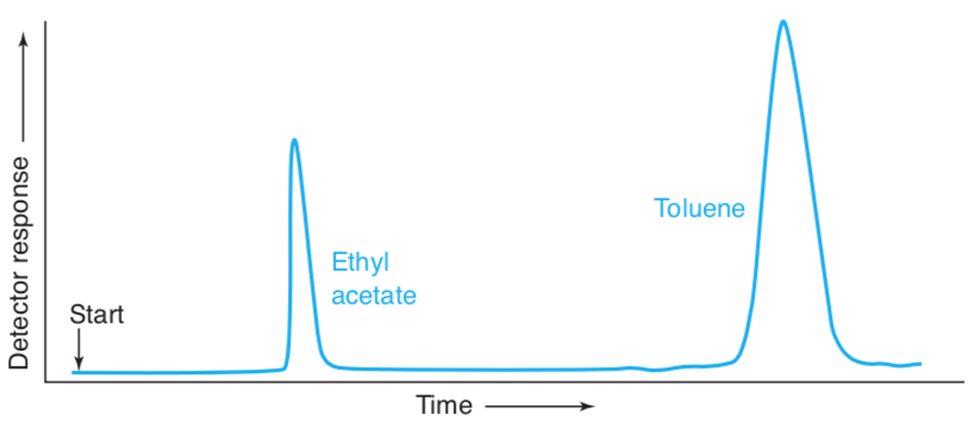
Chapter 23: Q35P (page 631)
Which column is more efficient: plate height = 0.1 mm or plate height = 1 mm?
Short Answer
The solution is
the column with a lower plate height of 0.1 mm would be preferred. Because there would be less band dispersion, it would be more efficient.