Chapter 23: Q26P (page 630)
The retention volume of a solute is 76.2mL for a column with and . Calculate the retention factor and the partition coefficient for this solute.
Short Answer
- Retention factor = k = 3.59
- Partition coefficient= K = 4.69
Learning Materials
Features
Discover
Chapter 23: Q26P (page 630)
The retention volume of a solute is 76.2mL for a column with and . Calculate the retention factor and the partition coefficient for this solute.
All the tools & learning materials you need for study success - in one app.
Get started for free
Consider the extraction of for which (a) Derive an expression for the fraction of metal ion extracted into the organic phase, in terms of the distribution coefficient and volumes of the two phases. (b) Prepare a graph of the percentage of metal ion extracted over the pH range 0 to 5.
The solute in Problem 23 - 8 is initially dissolved in 80.0 mL of water. It is then extracted six times with 10.0mL portions of chloroform. Find the fraction of solute remaining in the aqueous phase.
(a) Explain why the diffusion coefficient of is greater than that of
sucrose in Table 23-1.
(b) Make an order-of-magnitude estimate of the diffusion coefficient of water
vapor in air at 298K.
Match statements 1–5 with the band broadening terms in the second list.
1. Depends on radius of open tubular column.
2. Not present in an open tubular column.
3. Depends on length and radius of connecting tubing.
4. Increases with diffusion coefficient of solute.
5. Increases with thickness of stationary phase film.
A Multiple paths
B Longitudinal diffusion
Equilibration time in mobile phase
Equilibration time in stationary phase
EC Extra column band broadening
A Gas chromatogram of a mixture of toluene and ethyl acetate is shown here:
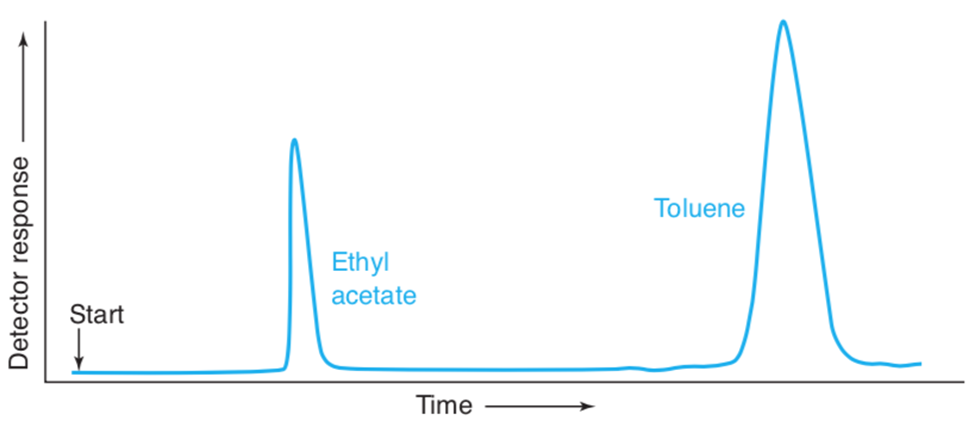
(a)Use the width of each peak (measured at the base)to calculate the number of theoretical plates in the column. Estimate all lengths to the nearest 0.1 mm.
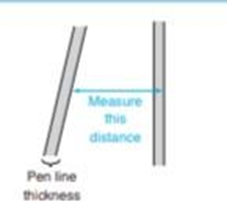
(b)Using the width of the toluene peak at its base, calculate the width expected at half height. Compare the measured and calculated values. When the thickness of the the line is significant relative to the length being measured, it is important to take the pen line width into account. You can measure from the edge of one line to the corresponding edge of the other line,as shown here.
What do you think about this solution?
We value your feedback to improve our textbook solutions.