Chapter 27: Q1TY (page 752)
How many grams ofwere in a sample that produced 1.000 g of
AgBr precipitate (FM 5 187.77)?
Short Answer
The number of bromide ions generated in a sample that can cause silver bromide precipitation is 0.4255g
Learning Materials
Features
Discover
Chapter 27: Q1TY (page 752)
How many grams ofwere in a sample that produced 1.000 g of
AgBr precipitate (FM 5 187.77)?
The number of bromide ions generated in a sample that can cause silver bromide precipitation is 0.4255g
All the tools & learning materials you need for study success - in one app.
Get started for free
Marie Curie dissolved 0.091 92 g of RaCl2 and treated it with excess AgNO3to precipitate 0.088 90 g of AgCl. In her time (1900), the atomic mass of Ag was known to be 107.8 and that of Cl
was 35.4. From these values, find the atomic mass of Ra that Marie Curie would have calculated.
We now examine some examples that illustrate how to relate the mass of a gravimetric precipitate to the quantity of the original analyte. The general approach is to relate the moles of product to the moles of reactant.
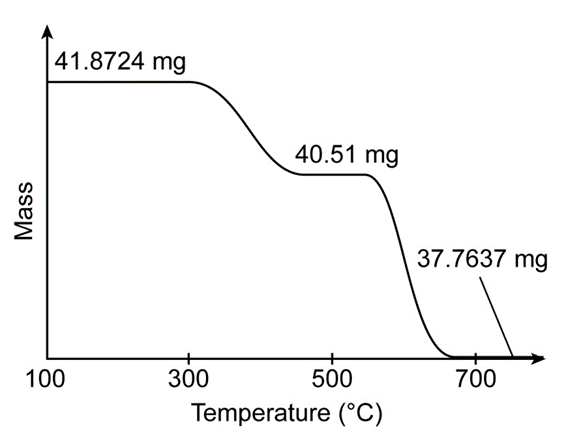
, has a perovskite structure (Box 19-1) with variable oxygen content. The figure shows the thermogravimetric curve observed when 41.8724m gwere heated in role="math" localid="1665035505591" does not react, but cobalt is reduced to Co(s)
(a) What is the oxidation state of cobalt in the ideal formula
(b) Write the reaction of role="math" localid="1665035588740" to produce
(c) If react completely, what will be the mass of product?
(d) Write the balanced reaction of role="math" localid="1665035782894" to produce react completely; what will be the mass of solid product? Your answer will be an expression with x in it.
(e) From the observed product mass , find x in Write the formula of the starting solid.
(f) In part (c), the mass lost from the ideal formula , is 4.0877 mg. What is the product nearrole="math" localid="1665035980286" where the mass is -40.51 mg?
What is the purpose of the and Cu in Figure 27-8?
Explain how the quartz crystal microbalance at the opening of Chapter 2 measures small masses
What do you think about this solution?
We value your feedback to improve our textbook solutions.