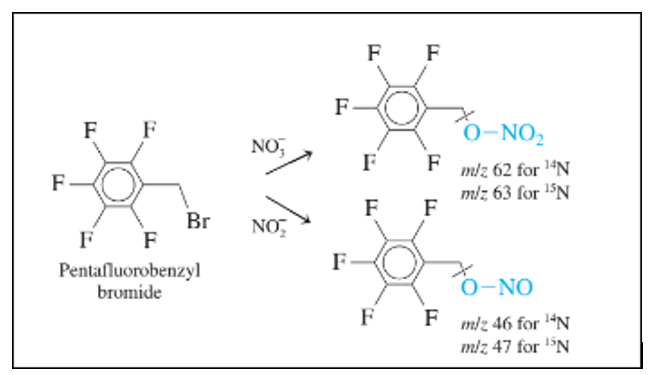
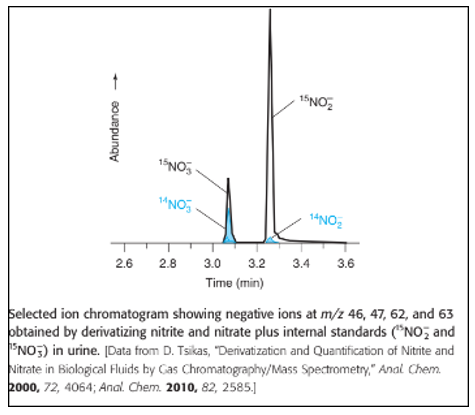
Chapter 24: Q30P (page 664)
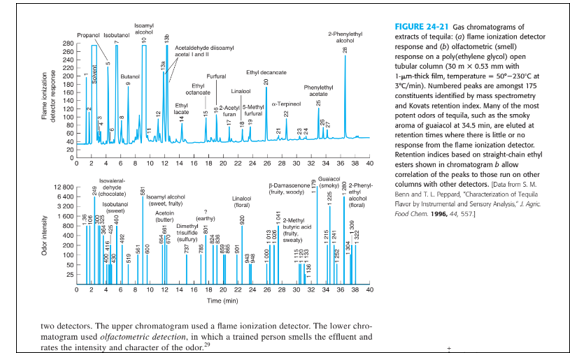
In the analysis of odorants in tequila in Figure 24-21, tequila was diluted with water and extracted four times with dichloromethane , b.p. Theofwas evaporated down toandof the extract was injected on-column onto a poly(ethylene glycol) open tubular column , film thickness ) initially at and then ramped to
(a) Why was the diluted tequila extracted four times with dichloromethane instead of once with a larger volume?
(b) Why was on-column injection used?
(c) Why was a poly(ethylene glycol) column chosen for this application?
(d) What was the phase ratio of the column?
(e) Why was a wide-bore 0.53-mm-diameter column chosen for this application?
Short Answer
(a). As a result, diluted tequila is extracted four times with dichloromethane rather than once at a higher volume.
(b).On-column injection minimises the likelihood of heat degradation as well as the odour of a larger number of molecules.
(c). The use of poly (ethylene glycol) in the column is due to the fact that poly (ethylene glycol) is a polar column, which is ideal for polar analytes.
(d).The column's phase ratio is 132.
(e).As a result, a wide-bore column with a diameter of enables for bigger injections of compounds, the scent of the components can be recognised during elution.