Chapter 3: Q15P (page 62)
Find the absolute and percent relative uncertainty and express each answer with a reasonable number of significant figures.
Short Answer
To calculate the absolute and percent relative uncertainty.
Learning Materials
Features
Discover
Chapter 3: Q15P (page 62)
Find the absolute and percent relative uncertainty and express each answer with a reasonable number of significant figures.
To calculate the absolute and percent relative uncertainty.
All the tools & learning materials you need for study success - in one app.
Get started for free
If uncertainty in pHis doubledto, what is the relative uncertainty in?
You have a stock solution certified by a manufacturer to contain . You would like to dilute it by a factor of 100 to obtain . Two possible methods of dilution are stated below. For each method, calculate the resulting uncertainty in concentration. Use manufacturer's tolerances in Tables 2-3 and 2-4 for uncertainties. Explain why one method is more precise than the other.
(a) Dilute 10.00mL up to 100mL with a transfer pipet and volumetric flask. Then take 10.00mL of the dilute solution and dilute it again to 100mL.
(b) Dilute 1.000mL up to 100mL with a transfer pipet and volumetric flask.
Cheryl, Cynthia, Carmen, and Chastity shot the following targets at Girl Scout camp. Match each target with the proper description.
(a) accurate and precise
(b) accurate but not precise
(c) precise but not accurate
(d) neither precise nor accurate
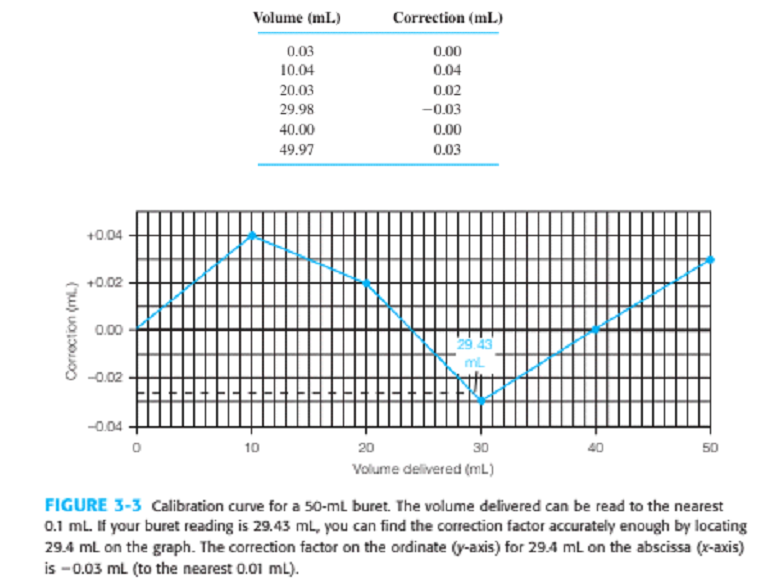
Controlling the appearance of a graph. Figure 3-3 requires gridlines to read buret corrections. In this exercise, you will format a graph so that it looks like Figure 3-3 . Follow the procedure in Section 2-11 to graph the data in the following table. For Excel 2007 or 2010, insert a Chart of the type Scatter with data points connected by straight lines. Delete the legend and title. With Chart Tools, Layout, Axis Titles, add labels for both axes. Click any number on the abscissa ( x axis) and go to Chart Tools, Format. In Format Selection, Axis Options, choose Minimum =0 , Maximum =50 , Major unit =10 , and Minor unit =1 . For Major tick mark type, select Outside. In Format Selection, Number, choose Number and set Decimal places =0. In a similar manner, set the ordinate ( y -axis) to run from -0.04 to +0.05 with a Major unit of 0.02 and a Minor unit of 0.01 and with Major tick marks Outside. To display gridlines, go to Chart Tools, Layout, and Gridlines. For Primary Horizontal Gridlines, select Major & Minor Gridlines. For Primary Vertical Gridlines, select Major & Minor Gridlines. To move axis labels from the middle of the chart to the bottom, click a number on the y axis (not the x axis) and select Chart Tools, Layout, Format Selection. In Axis Options, choose Horizontal axis crosses Axis value and type in -0.04 . Close the Format Axis window and your graph should look like Figure 3-3 .
Compute the molecular mass and its uncertainty for . What is the percent relative uncertainty in molecular mass?
What do you think about this solution?
We value your feedback to improve our textbook solutions.