Chapter 3: Q5TY (page 58)
If uncertainty in pHis doubledto, what is the relative uncertainty in?
Short Answer
An uncertainty of 0.06 (doubled) in pH gives an uncertainty of 14% in
Learning Materials
Features
Discover
Chapter 3: Q5TY (page 58)
If uncertainty in pHis doubledto, what is the relative uncertainty in?
An uncertainty of 0.06 (doubled) in pH gives an uncertainty of 14% in
All the tools & learning materials you need for study success - in one app.
Get started for free
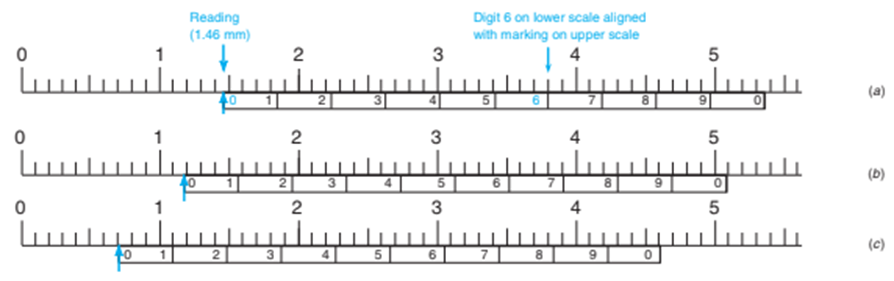
Vernier scale. The figure below shows a scale found on instruments such as a micrometer caliper used for accurately measuring dimensions of objects. The lower scale slides along the upper scale and is used to interpolate between the markings on the upper scale. In (a), the reading (at the left-hand 0 of the lower scale) is between 1.4and 1.5on the upper scale. To find the exact reading, observe which mark on the lower scale is aligned with a mark on the upper scale. Because theon the lower scale is aligned with the upper scale, the correct reading is 1.46. Write the correct readings in (b) and (c) and indicate how many significant figures are in each reading.
(a) Show that (b) To prepare a solution of NaCI , you weigh out 2.634(60.002)g and dissolve it in a volumetric flask whose volume is 100.00(60.08)mL Express the molarity of the solution, along with its uncertainty, with an appropriate number of digits.
Why do we use quotation marks around the word true in the statement that accuracy refers to how close a measured value is to the "true" value?
Find the absolute and percent relative uncertainty and express each answer with a reasonable number of significant figures.
Round each number as indicated:
What do you think about this solution?
We value your feedback to improve our textbook solutions.