Chapter 17: Q40P (page 431)
What is the purpose of the Nafion the membrane in Figure 17-33
Short Answer
The benefits of nafion membrane in given figure has to be given.
Learning Materials
Features
Discover
Chapter 17: Q40P (page 431)
What is the purpose of the Nafion the membrane in Figure 17-33
The benefits of nafion membrane in given figure has to be given.
All the tools & learning materials you need for study success - in one app.
Get started for free
What is a Clark electrode, and how does it work?
Explain how the endpoint is detected in a Karl Fischer titration in Figure 17-35.
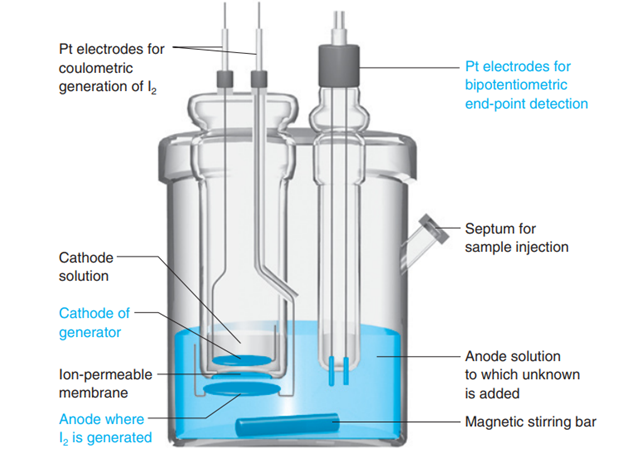
Explain how amperometric end-point detection in Figure17-9 operates.
Peak current and scan rate (v) are listed for cyclic voltammetry of the reversible reaction Fe( II ) Fe(III ) of a water-soluble ferrocene derivative in 0.1MNaCLIf a graph of versusgives a straight line, then the reaction is diffusion controlled. Prepare such a graph and use it to find the diffusion coefficient of the reactant from Equation17 - 21for this one-electron oxidation. The area of the working electrode is, and the concentration of reactant is 1.00mM.
Chlorine has been used for decades to disinfect drinking water. An undesirable side effect of this treatment is reaction with organic impurities to create organochlorine compounds, some of which could be toxic. Monitoring total organic halide (designated TOX) is required for many water providers. A standard procedure for TOX is to pass water through activated charcoal, which adsorbs organic compounds. Then the charcoal is combusted to liberate hydrogen halides:
HX is absorbed into aqueous solution and measured by coulometric titration with a silver anode:
When 1.00 L of drinking water was analyzed, a current of 4.23 mA was required for 387 s. A blank prepared by oxidizing charcoal required 6 s at 4.23 mA. Express the TOX of the drinking water as mol halogen/L. If all halogen is chlorine, express the TOX as .
What do you think about this solution?
We value your feedback to improve our textbook solutions.