Chapter 17: Q29P (page 430)
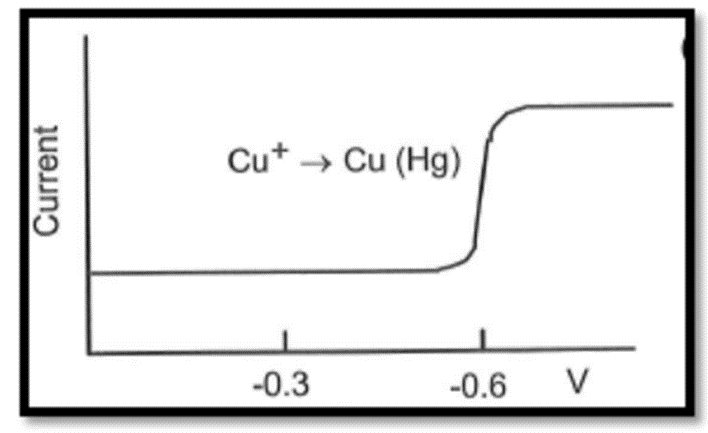
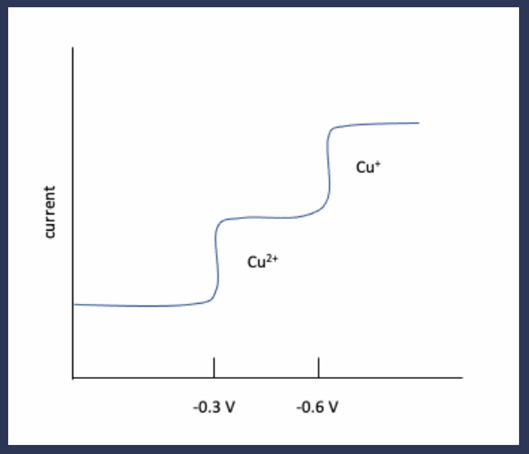
In solution,is reduced tonear -0.3V(versus S.C.E.), andis reduced tonear 0.6V.
(a) Sketch a qualitative sampled current polarogram for a solution of.
(b) Sketch a polarogram for a solution of.
(c) Suppose that Pt, instead of Hg, were used as the working electrode. Which, if any, reduction potential would you expect to change?
Short Answer
a) The reduction of copper ion leads to copper-amalgam
b) The reduction of forms and further reduction with increase in voltage leads to copper-amalgam.
c) The reduction potential for the reduction reaction of copper ion to copper will obviously change because the working electrode is changed to platinum.