Chapter 17: Q7 P (page 427)
The Weston cell is a stable voltage standard formerly used in potentiometers. (The potentiometer compares an unknown voltage with that of the standard. In contrast with the conditions of this problem, very little current may be drawn from the cell if it is to be a voltage standard.)
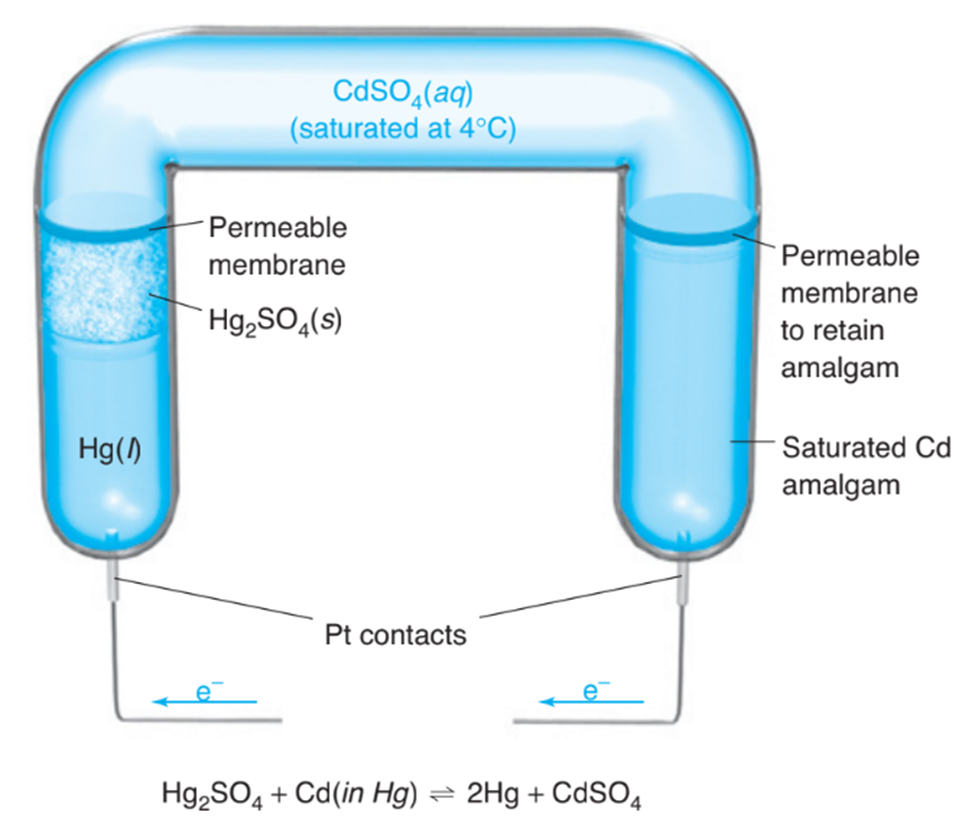
How much work (J)can be done by the Weston cell if the voltage is 1.02 V and1.00mLof Hg(density = 13.52g/mL) is deposited?
(b) If the cell passes current through a resistor that dissipates heat at a rate of0.209 J/min, how many grams of are oxidized each hour? (This question is not meant to be consistent with part (a). The voltage is no longer 1.02volts.)
Short Answer
(a) The work is W = 6638.1J
(b) The mass of Cd is m(Cd) = 0.01237g

