Chapter 17: Q1Q (page 412)
Is B-doped diamond p-type or n-type? Consult Figure 15-37
Short Answer
Diamond doped with boron is P-type semiconductor
Learning Materials
Features
Discover
Chapter 17: Q1Q (page 412)
Is B-doped diamond p-type or n-type? Consult Figure 15-37
Diamond doped with boron is P-type semiconductor
All the tools & learning materials you need for study success - in one app.
Get started for free
Coulometric titration of sulfite in wine. Sulfur dioxide is added to many foods as a preservative. In aqueous solution, the following species are in equilibrium:
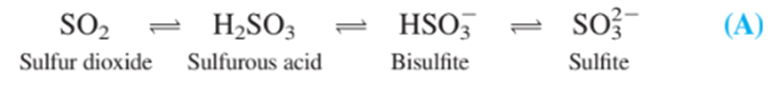
Bisulfite reacts with aldehydes in food near neutral pH:
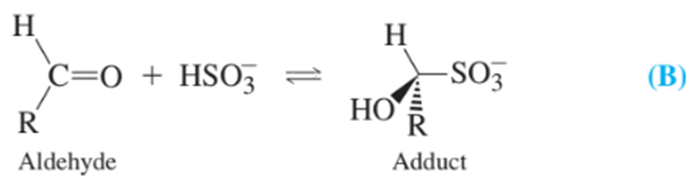
Sulfite is released from the adduct in 2MNaOH and can be analyzed by its reaction with to give and sulfate. Excess must be present for quantitative reaction.
Here is a coulometric procedure for analysis of total sulfite in white wine. Total sulfite means all species in Reaction and the adduct in Reaction . We use white wine so that we can see the color of a starch-iodine end point.
1. Mix 9.00 mL of wine plus 0.8gNaOH and dilute to 10.00mL. The releases sulfite from its organic adducts.
2. Generate at the working electrode (the anode) by passing a known current for a known time through the cell in Figure 17 - 10. The cell containsofacetate buffer () plus. In the cathode compartment, is reduced to . The frit retards diffusion of into the main compartment, where it would react with to give.
3. Generate at the anode with a current of for.
4. Inject 2.000mL of the wine/ solution into the cell, where the sulfite reacts with leaving excess.
5. Add of thiosulfate to consume by Reaction and leave excess thiosulfate.
6. Add starch indicator to the cell and generate fresh with a constant current of 10.0mA. A time of 131s was required to consume excess thiosulfate and reach the starch end point.
(a) In what pH range is each form of sulfurous acid predominant?
(b) Write balanced half-reactions for the anode and cathode.
(c) At pH 3.7, the dominant form of sulfurous acid isand the dominant form of sulfuric acid is . Write balanced reactions between andand between and thiosulfate.
(d) Find the concentration of total sulfite in undiluted wine.
The Weston cell is a stable voltage standard formerly used in potentiometers. (The potentiometer compares an unknown voltage with that of the standard. In contrast with the conditions of this problem, very little current may be drawn from the cell if it is to be a voltage standard.)
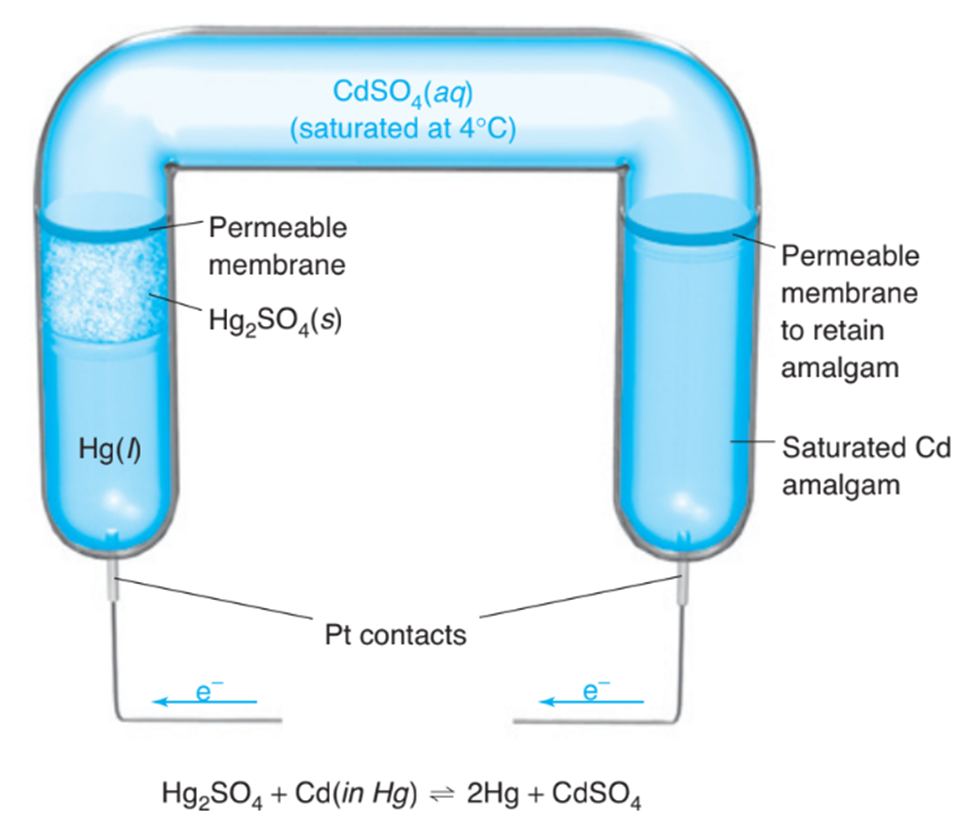
How much work (J)can be done by the Weston cell if the voltage is 1.02 V and1.00mLof Hg(density = 13.52g/mL) is deposited?
(b) If the cell passes current through a resistor that dissipates heat at a rate of0.209 J/min, how many grams of are oxidized each hour? (This question is not meant to be consistent with part (a). The voltage is no longer 1.02volts.)
(a) which voltage,orin the diagram is constant in controlled-potential electrolysis? Which are the working, auxiliary, and reference electrodes in the diagram?
(b) Explain how the Luggin capillary in Figure 17-4 measures the electric potential at the opening of the capillary.
Chlorine has been used for decades to disinfect drinking water. An undesirable side effect of this treatment is reaction with organic impurities to create organochlorine compounds, some of which could be toxic. Monitoring total organic halide (designated TOX) is required for many water providers. A standard procedure for TOX is to pass water through activated charcoal, which adsorbs organic compounds. Then the charcoal is combusted to liberate hydrogen halides:
HX is absorbed into aqueous solution and measured by coulometric titration with a silver anode:
When 1.00 L of drinking water was analyzed, a current of 4.23 mA was required for 387 s. A blank prepared by oxidizing charcoal required 6 s at 4.23 mA. Express the TOX of the drinking water as mol halogen/L. If all halogen is chlorine, express the TOX as .
Propagation of uncertainty. In an extremely accurate measurement of the Faraday constant, a pure silver anode was oxidized to Ag1 with a constant current of 0.2036390(60.0000004)A for18000.075(60.010s to give a mass loss of4.097900(60.0000003) g from the anode. Given that the atomic mass of Ag is 107.8682 (60.0002), find the value of the Faraday constant and its uncertainty.
What do you think about this solution?
We value your feedback to improve our textbook solutions.