Chapter 6: Q25P (page 142)
Given the following equilibria, calculate the concentration of
each zinc species in a solution saturated withand containing
at a fixed concentration of.
Learning Materials
Features
Discover
Chapter 6: Q25P (page 142)
Given the following equilibria, calculate the concentration of
each zinc species in a solution saturated withand containing
at a fixed concentration of.
All the tools & learning materials you need for study success - in one app.
Get started for free
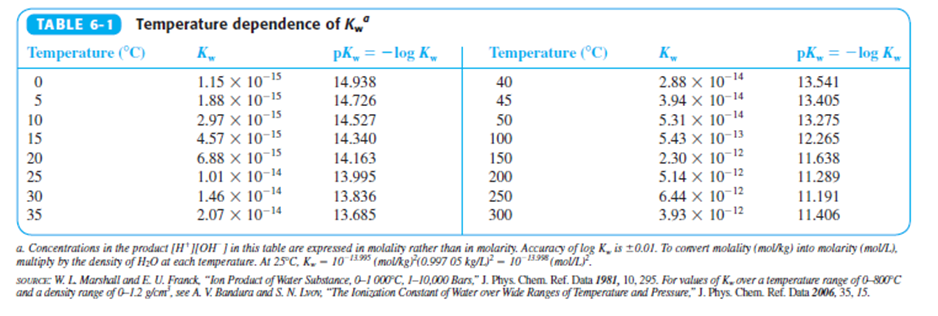
Use Le Châtelier’s principle and Kw in Table 6-1 to decide whether the autoprotolysis of water is endothermic or exothermic at 250C; (b)1000C; (c)3000C.
for chloroacetic acid is Find for chloroacetate ion.
Identify the Lewis acids in the following reactions:
Find and in a saturated solution of with
The equilibrium constant for the reaction of isat and at .
(a) Assuming and are constant in the interval (probably a good assumption for small ), use Equation 6-9 to find for the reaction in this temperature range.
(b) Describe how Equation 6-9 could be used to make a linear graph to determine , if and were constant over some temperature range
What do you think about this solution?
We value your feedback to improve our textbook solutions.