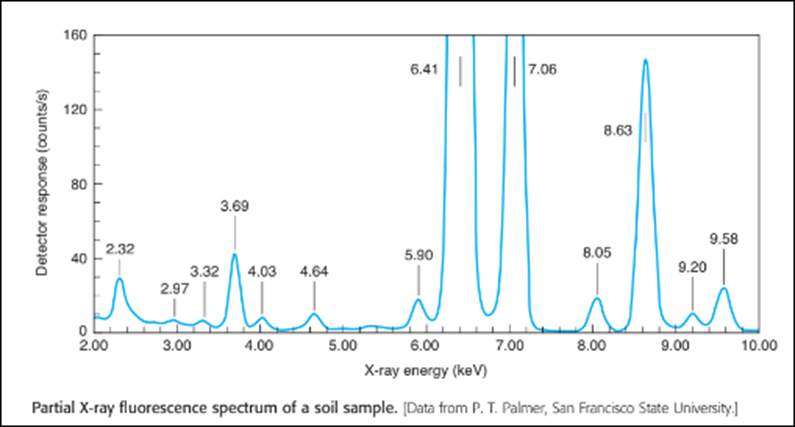
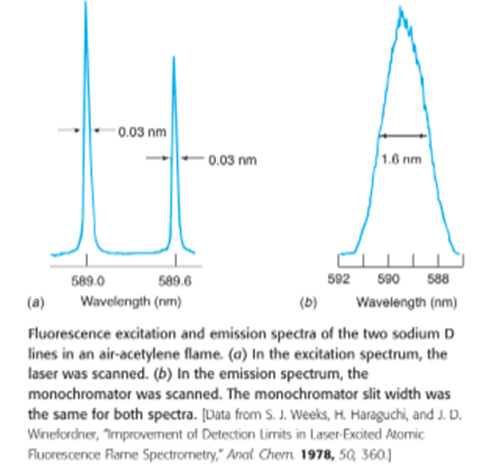
Chapter 21: QB E (page 554)
Mn was used as an internal standard for measuring Fe by atomic absorption. A standard mixture containing andgave a quotient (Fe signal/Mn signal) = 1.05 / 1.00. A mixture with a volume of 6.00 mLwas prepared by mixing 5.00 mLof unknown Fe solution with 1.00mLcontaining. The absorbance of this mixture at the Mn wavelength was 0.128, and the absorbance at the Fe wavelength was 0.185. Find the molarity of the unknown Fe solution.
Short Answer
The molarity of a Fe solution with an unknown molarity is