Chapter 21: QE E (page 555)
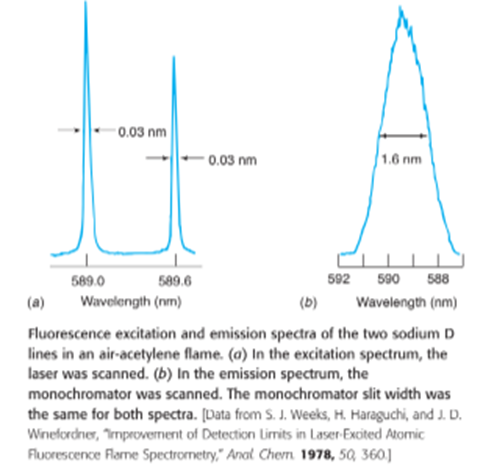
Assign as many of the peaks as you can in the X-ray fluorescence spectrum shown here. For each peak, state where the peak should appear and state whether there is a plausible peak at the position. There must be a for every , but the localid="1663657296432" intensity should beof theintensity.
Short Answer
The following are some examples of probable peak assignments,