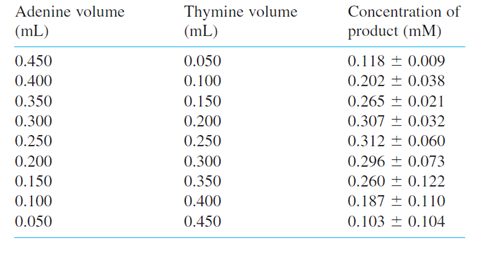
Chapter 19: Q14P (page 487)
Now we use Solver to find Kfor the previous problem. The only absorbing species at 332 nmis the complex, so, from Beer’s law is either free or bound in the complex,so There is a huge excess of mesitylene, so
The spreadsheet shows some of the data. You will need to use all the data. Column A contains [mesitylene] and column B contains . Column C lists the measured absorbance. Guessa value of the molar absorptivity of the complex,Then compute the concentration of the complex in column D. The equilibrium constant in column
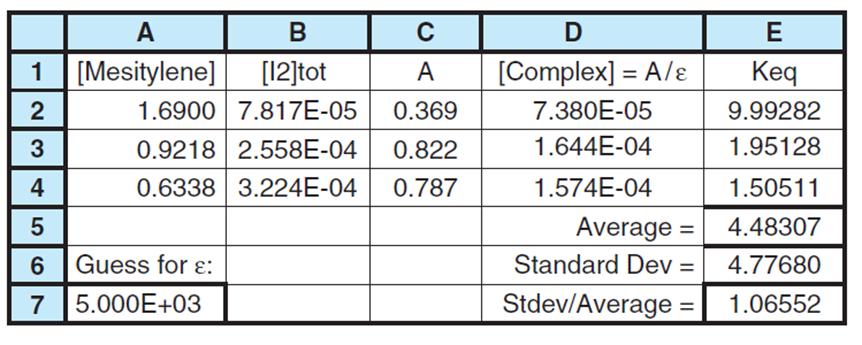
should we minimize with Solver? We want to varyεin cell A7 until the values of Kin column E are as constant as possible. We would like to minimize a function like , where Kiis the value in each line of the table and Kaverage is the average of all computed values. The problem with is that we can minimize this function simply by making very small, but not necessarily constant. What we really want is for all the to be clustered around the mean value. A good way to do this is to minimize the relative standard deviationof the K, which is (standard deviation)/average. In cell E5we compute the average value of Kand in cell E6the standard deviation. Cell E7contains the relative standard deviation. Use Solver to minimize cell E7by varying cell A7. Compare your answer with that of Problem 19-13.
Short Answer
The equilibrium constant is 0.464 and molar absorptivity is .