Chapter 11: Q2TY (page 245)
Verify the derivative in cell D7 of Figure 11-6.
Short Answer
The value 0.226 in cell D7 is verified.
Learning Materials
Features
Discover
Chapter 11: Q2TY (page 245)
Verify the derivative in cell D7 of Figure 11-6.
The value 0.226 in cell D7 is verified.
All the tools & learning materials you need for study success - in one app.
Get started for free
Titrating weak acid with weak base.
(a) Prepare a family of graphs for the titration of 50.0 mL of 0.020 0 M HA (pKa = 4.00) with 0.100 M B (pKb = 3.00, 6.00, and 9.00).
(b) Write the acid-base reaction that occurs when acetic acid and sodium benzoate (the salt of benzoic acid) are mixed, and find the equilibrium constant for the reaction. Find the pH of a solution prepared by mixing 212 mL of 0.200 M acetic acid with 325 mL of 0.050 0 M sodium benzoate.
Pyridine is half protonated in aqueous phosphate buffer at pH 5.2. If you mix 45 mL of phosphate buffer with 55 mL of methanol, the buffer must have a pH of 3.2 to half protonate pyridine. Suggest a reason why.
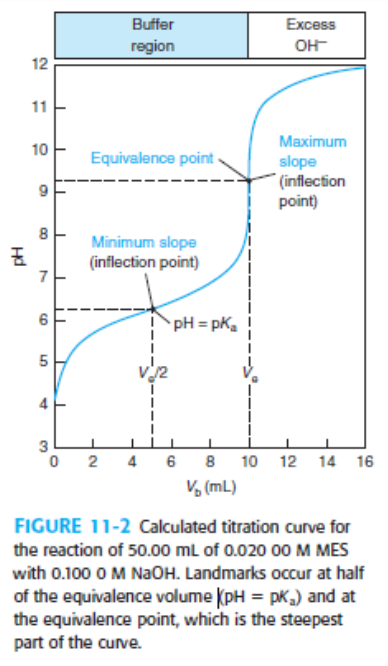
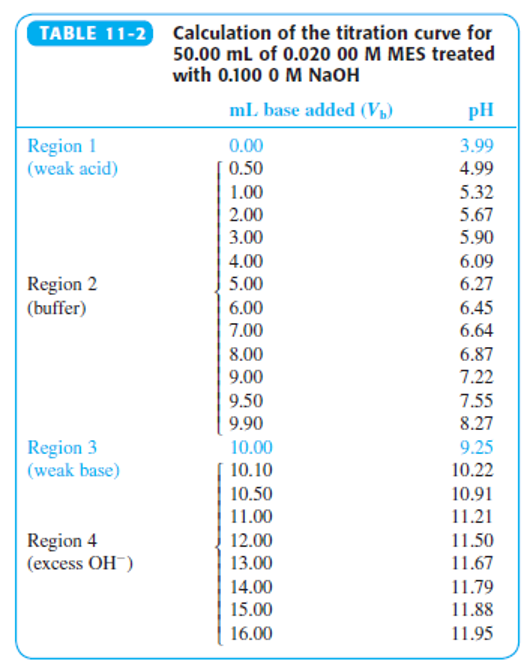
Indicator error. Consider the titration in Figure 11-2 in which the equivalence-point pH in Table 11-2 is 9.25 at a volume of 10.00 mL.
(a) Suppose you used the yellow-to-blue transition of thymol blue indicator to find the end point. According to Table 11-3, the last trace of green disappears near pH 9.6. What volume of base is required to reach pH 9.6? The difference between this volume an 10 mL is the indicator error.
(b) If you used cresol red, with a color change at pH 8.8, what would be the indicator error?
Titration on Diprotic Systems
How many grams of dipotassium oxalate ( ) should be added to of to give a of when the solution is diluted to ?
Titration on Diprotic Systems
. Calculate the whenrole="math" localid="1655019122863" of aminophenol have been titrated with role="math" localid="1655019100358" of .
What do you think about this solution?
We value your feedback to improve our textbook solutions.