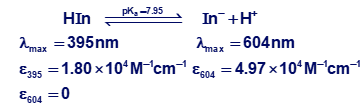Chapter 11: QJE (page 259)
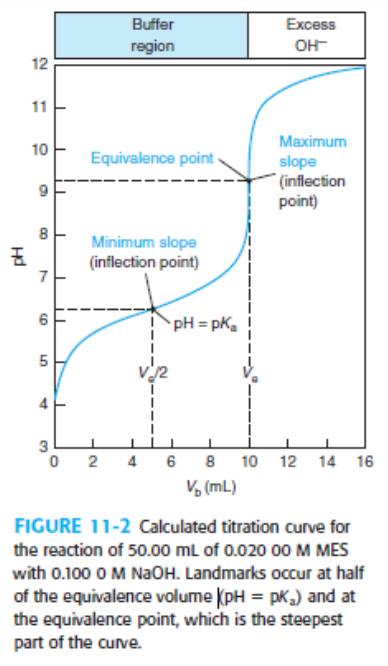
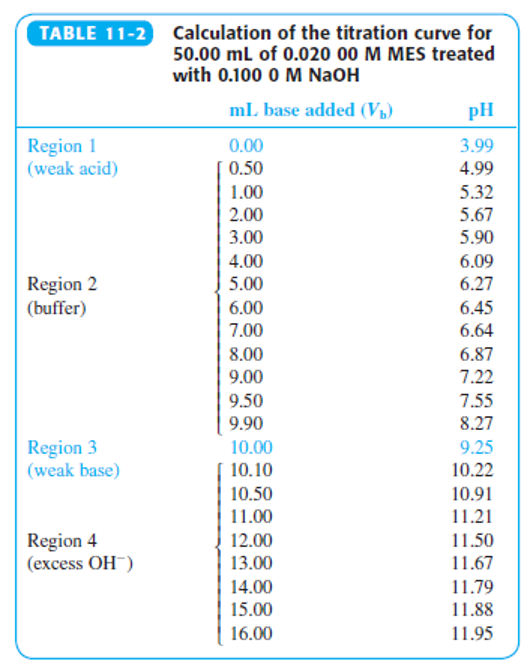
Indicator error. Consider the titration in Figure 11-2 in which the equivalence-point pH in Table 11-2 is 9.25 at a volume of 10.00 mL.
(a) Suppose you used the yellow-to-blue transition of thymol blue indicator to find the end point. According to Table 11-3, the last trace of green disappears near pH 9.6. What volume of base is required to reach pH 9.6? The difference between this volume an 10 mL is the indicator error.
(b) If you used cresol red, with a color change at pH 8.8, what would be the indicator error?
Short Answer
(a) The volume of base is required to reach pH is .
(b) The indicator error is .