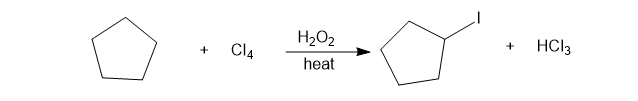
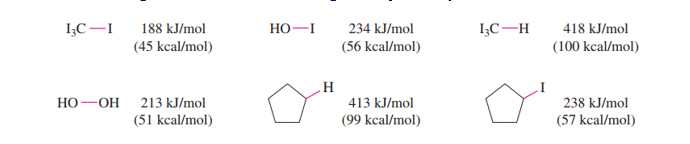
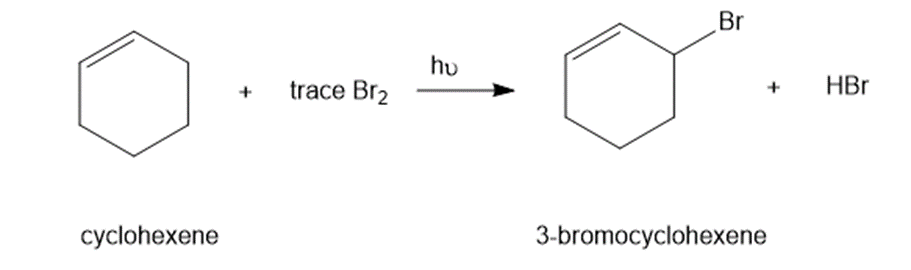
Chapter 4: Q12P (page 206)
Under certain conditions, the bromination of cyclohexene follows an unusual rate law:
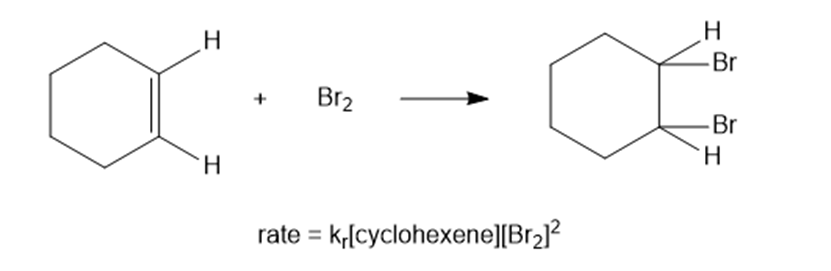
(a) What is the kinetic order with respect to cyclohexene?
(b) What is the kinetic order with respect to bromine?
(c) What is the kinetic order overall?
Short Answer
(a) First order.
(b) Second order.
(c) Third order.