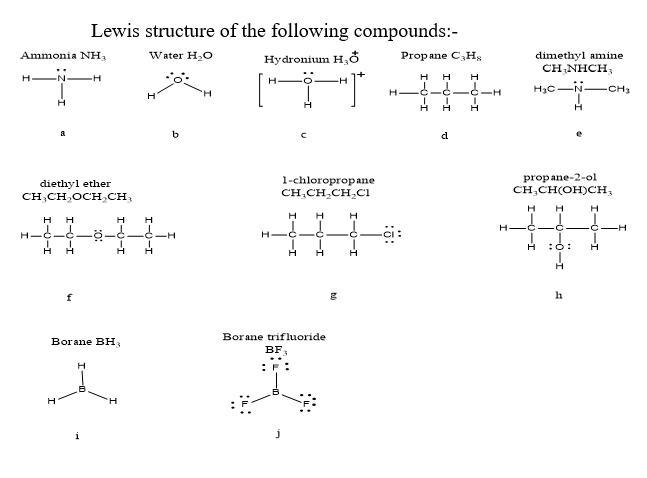
Chapter 1: Q2P (page 60)
Draw Lewis structures for the following compounds.
- ammonia, NH3
- water, H2O
- hydronium, H3O+
- propane, C 3H8
- dimethylamine, CH3NHCH3
- diethyl ether, CH3CH2OCH2CH3
- 1-chloropropane, CH3CH2CH2Cl
- propan-2-ol, CH3CH(OH)CH3
- borane, BH3
- boron trifluoride, BF3
Explain what is unusual about the bonding in the compounds in parts (i) and (j).
Short Answer
Lewis structures for the compounds are as follows.