Chapter 17: Q54P (page 907)
Predict the major products of bromination of the following compounds, using Br2 and FeBr3 in the dark.
(a)
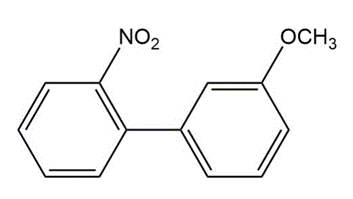
(b)
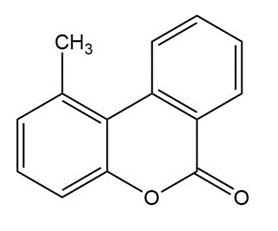
(c)
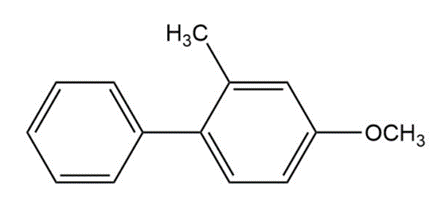
Short Answer
(a)
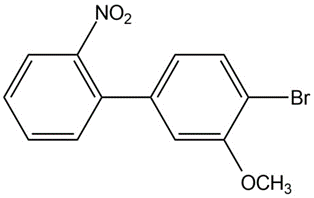
(b)
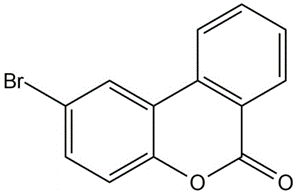
(c)
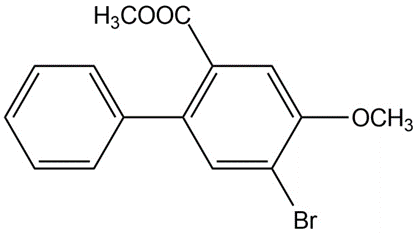
Learning Materials
Features
Discover
Chapter 17: Q54P (page 907)
Predict the major products of bromination of the following compounds, using Br2 and FeBr3 in the dark.
(a)

(b)

(c)

(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Propose a mechanism for the bromination of ethyl benzene shown above.
Indane can undergo free-radical chlorination at any of the alkyl positions on the aliphatic ring.
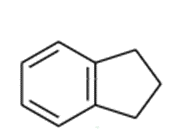
indane
Propose products (if any) and mechanisms for the following AICI3-catalyzed reactions:
⦁ chlorocyclohexane with benzene
⦁ methyl chloride with anisole
⦁ 3-chloro-2,2-dimethylbutane with isopropylbenzene
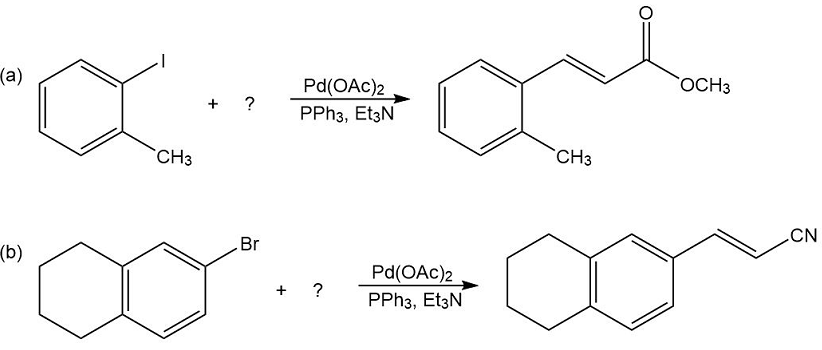
What substituted alkene would you use in the Heck reaction to make the following products?
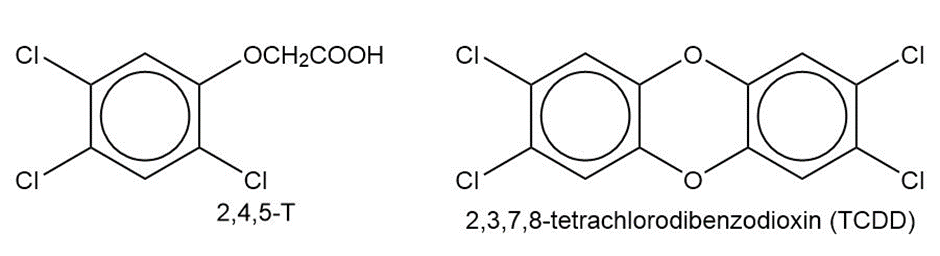
In Chapter 14, we saw that Agent Orange contains (2,4,5-trichlorophenoxy) acetic acid, called 2,4,5-T. This compound is synthesized by the partial reaction of 1,2,4,5-tetrachlorobenzene with sodium hydroxide, followed by a reaction with sodium chloroacetate, ClCH2CO2Na.
(a) Draw the structures of these compounds, and write equations for these reactions.
(b) One of the impurities in the Agent Orange used in Vietnam was 2,3,7,8-tetrachlorodibenzodioxin (2,3,7,8-TCDD), often incorrectly called “dioxin.” Propose a mechanism to show how 2,3,7,8-TCDD is formed in the synthesis of 2,4,5-T.
(c) Show how the TCDD contamination might be eliminated, both after the first step and on completion of the synthesis
What do you think about this solution?
We value your feedback to improve our textbook solutions.