Chapter 17: Q42P (page 845)
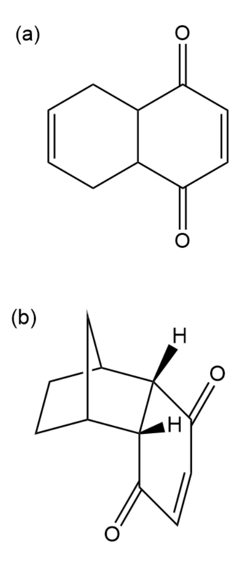
1,4-Benzoquinone is a good Diels–Alder dienophile. Predict the products of its reaction with
(a) buta-1,3-diene
(b) cyclopenta-1,3-diene
Short Answer
Learning Materials
Features
Discover
Chapter 17: Q42P (page 845)
1,4-Benzoquinone is a good Diels–Alder dienophile. Predict the products of its reaction with
(a) buta-1,3-diene
(b) cyclopenta-1,3-diene

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would use Suzuki reactions to synthesize these products from the indicated starting materials. You may use any additional reagents you need.
Which reactions will produce the desired product in good yield? You may assume that aluminum chloride is added as a catalyst in each case. For the reactions that will not give a good yield of the desired product, predict the major products.
Reagents Desired Product
(a) benzene + n-butyl bromide n-butylbenzene
(b) ethylbenzene + tert-butyl chloride p-ethyl-tert-butylbenzene
(c) bromobenzene + ethyl chloride p-bromoethylbenzene
(d) benzamide (PhCONH3) + CH3 CH2 CI p-ethylbenzamide
(e) toluene + HNO3, H2SO4 , heat 2,4,6-trinitrotoluene (TNT)
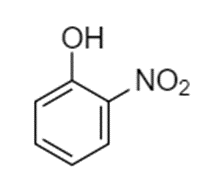
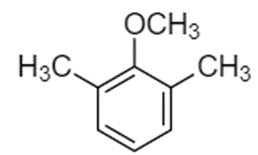
Predict the site(s) of electrophilic attack on these compounds.
(a)
(b)
Triphenylmethanol is insoluble in water, but when it is treated with concentrated sulfuric acid, a bright yellow solution results. As this yellow solution is diluted with water, its color disappears and a precipitate of triphenylmethanol reappears. Suggest a structure for the bright yellow species, and explain this unusual behavior.
Indane can undergo free-radical chlorination at any of the alkyl positions on the aliphatic ring.
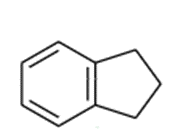
indane
What do you think about this solution?
We value your feedback to improve our textbook solutions.