Chapter 8: Q60P. (page 460)
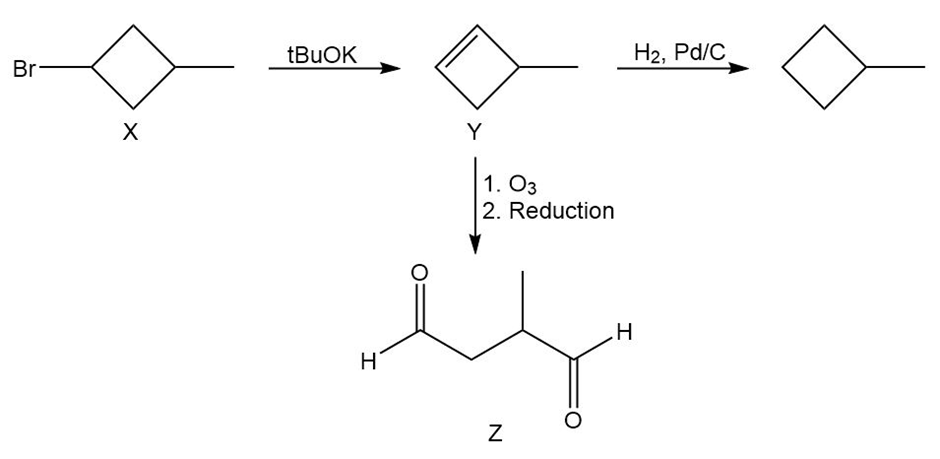
Unknown X, C5 H9 Br, does not react with bromine or with dilute KMnO4. Upon treatment with potassium tert-butoxide, X gives only one product, Y, C5 H8. Unlike X, Y decolorizes bromine and changes KMnO4 from purple to brown. Catalytic hydrogenation of Y gives methylcyclobutane. Ozonolysis–reduction of Y gives dialdehyde Z, C5 H8 O2. Propose consistent structures for X, Y, and Z. Is there any aspect of the structure of X that is still unknown?
Short Answer
Proposed structures with reactions are shown below.