Chapter 8: Q19Q (page 395)
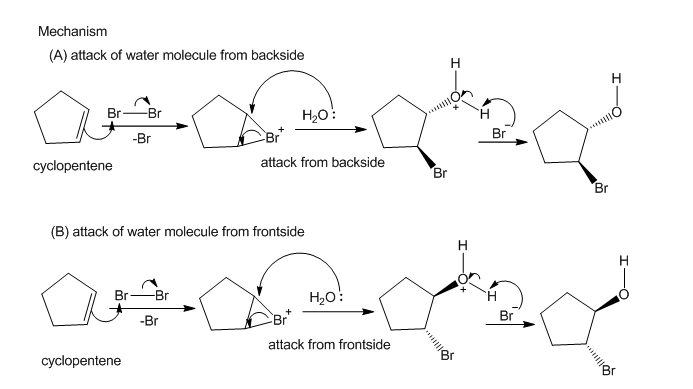
Propose a mechanism for the addition of bromine water to cyclopentene, being carefulto show why the trans product results and how both enantiomers are formed.
Short Answer
Learning Materials
Features
Discover
Chapter 8: Q19Q (page 395)
Propose a mechanism for the addition of bromine water to cyclopentene, being carefulto show why the trans product results and how both enantiomers are formed.

All the tools & learning materials you need for study success - in one app.
Get started for free
The chiral BINAP ligand shown in figure 8-8 contains no asymmetric carbon atoms. Explain how this ligand is chiral.
Predict the carbenoid addition products of the following reactions.
(a) trans-hex-3-ene+ CH2I2, Zn(Cu)
(b) ciz-hept-2-ene+ CH2I2, Zn(Cu)
Propose a mechanism to show how 3,3-dimethylbut-1-ene reacts with dilute aqueous H2SO4to give 2,3-dimethylbutan-2-ol and a small amount of 2,3-dimethylbut-2-ene.
One of the principal components of lemongrass oil is limonene, . When limonene is treated with excess hydrogen and a platinum catalyst, the product is an alkane of formula . What can you conclude about the structure of limonene?
Question: Show how you would accomplish the following synthetic conversions.
What do you think about this solution?
We value your feedback to improve our textbook solutions.