Chapter 13: Q56P (page 707)
Show how you would distinguish among the following threecompounds
Short Answer
Learning Materials
Features
Discover
Chapter 13: Q56P (page 707)
Show how you would distinguish among the following threecompounds


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: An unknown compound has the molecular formula C9H11Br. Its proton NMR spectrum shows the following absorptions: singlet, d7.1, integral 44 mm singlet, d2.3, integral 130 mm singlet, d2.2, integral 67 mm Propose a structure for this compound.
A small pilot was adding bromine across the double bond of but-2-ene to make 2,3-dibromobutane. A controller malfunction allowed the reaction temperature to rise beyond safe limits. A careful distillation of the product showed that several impurities had formed, including the one having the NMR spectra that appear below. Determine its structure and assign the peaks to the protons in your structure.
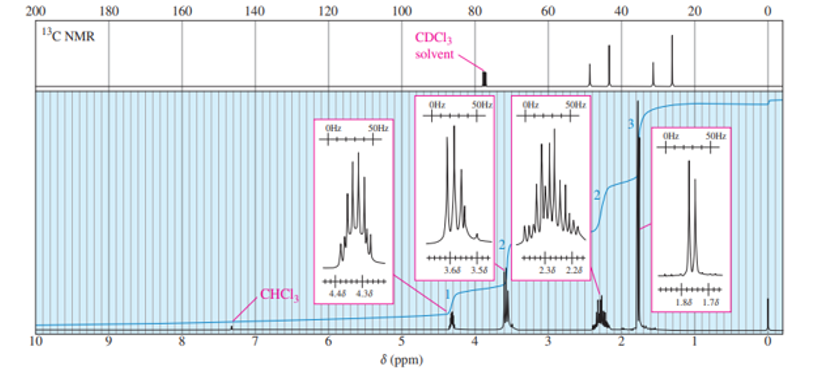
A new chemist moved into an industrial lab where work was being done on oxygenated gasoline additives. Among the additives that had been tested, she found an old bottle containing a clear, pleasant-smelling liquid that was missing its label. She took the quick NMR spectrum shown and was able to determine the identity of the compound without any additional information. Propose a structure and assign the peaks. (Hint: This is a very pure sample.)
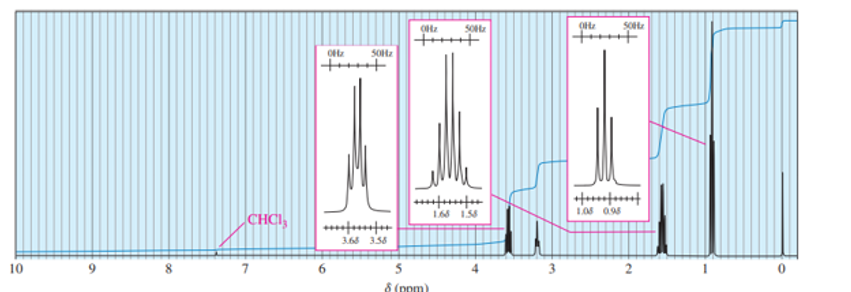
Draw the expected broadband-decoupled 13C NMR spectra of the following compounds. Use Figure 13-41 (page 686) to estimate the chemical shift.
Each of these four structures has molecular formula . Match the structure with its characteristic proton NMR signals. (Not all of the signals are listed in each case.)
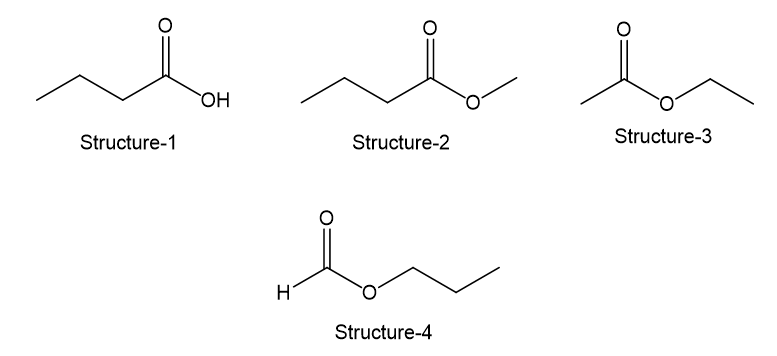
(a) Sharpsinglet atδ8.0 andtriplet atδ4.0
(b) Sharpsinglet atδ2.0 andquartet atδ4.1
(c) Sharpsinglet atδ3.7 andquartet atδ2.3
(d) Broadsinglet atδ11.5 andtriplet atδ2.3
What do you think about this solution?
We value your feedback to improve our textbook solutions.