Chapter 18: Q48P (page 969)
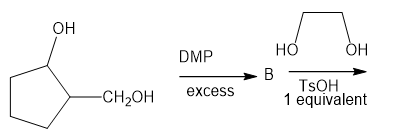
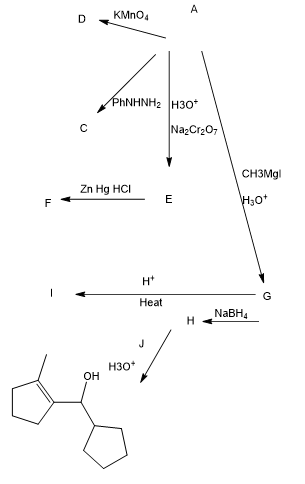
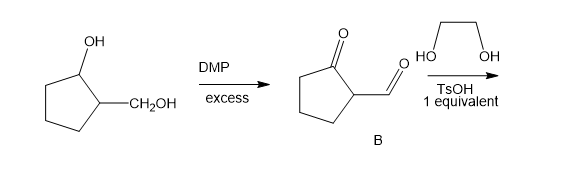
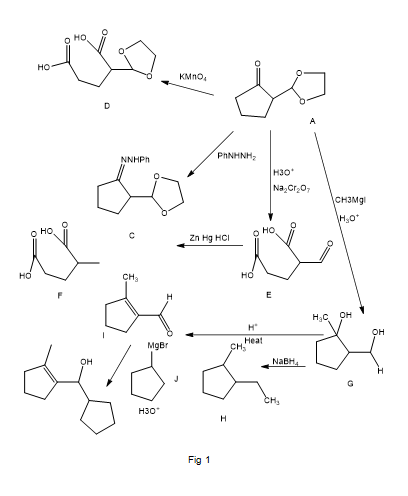
The following road-map problem centers on the structure and properties of A, a key intermediate in these reactions. Give structures for compounds A through J.
Short Answer
Learning Materials
Features
Discover
Chapter 18: Q48P (page 969)
The following road-map problem centers on the structure and properties of A, a key intermediate in these reactions. Give structures for compounds A through J.




All the tools & learning materials you need for study success - in one app.
Get started for free
Two structures for the sugar glucose are shown on page 950. Interconversion of the open-chain and cyclic hemiacetal forms is catalyzed by either acid or base.
(a) Propose a mechanism for the cyclization, assuming a trace of acid is present.
(b) The cyclic hemiacetal is more stable than the open-chain form, so very little of the open-chain form is present at equilibrium. Will an aqueous solution of glucose reduce Tollens reagent and give a positive Tollens test? Explain.
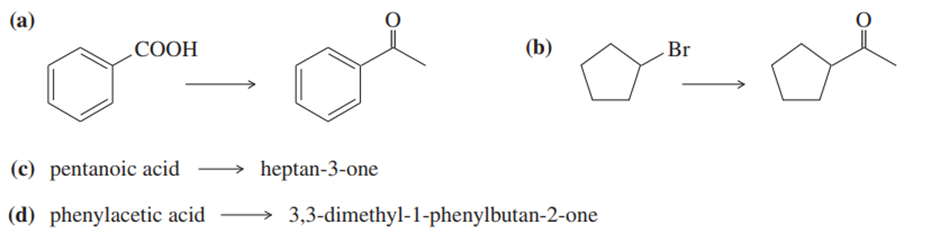
Show how you would accomplish the following synthetic conversions by adding an organolithium reagent to an acid.
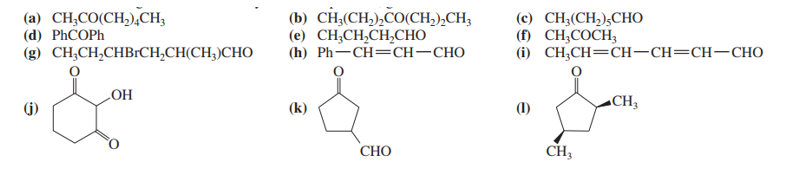
Name the following ketones and aldehydes. When possible, give both a common name and an IUPAC name.
A compound of formula C6H10O2 shows only two absorptions in proton NMR: a singlet at 2.67 ppm and singlet at 2.15 ppm. These absorptions have areas in the ratio 2:3. The IR spectrum shows strong absorption at 1708 cm-1. Propose structure of compound.
The Wittig reaction is useful for placing double bonds in less stable positions. For example, the following transformation is easily accomplished using a Wittig reaction
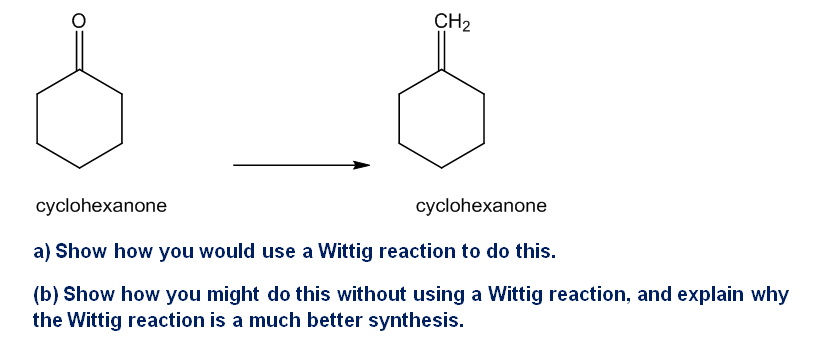
What do you think about this solution?
We value your feedback to improve our textbook solutions.