Chapter 15: Q7P (page 761)
Propose a mechanism for each reaction, showing explicitly how the observed mixtures of products are formed.
a)
b)c)d)
e)
Short Answer
a)
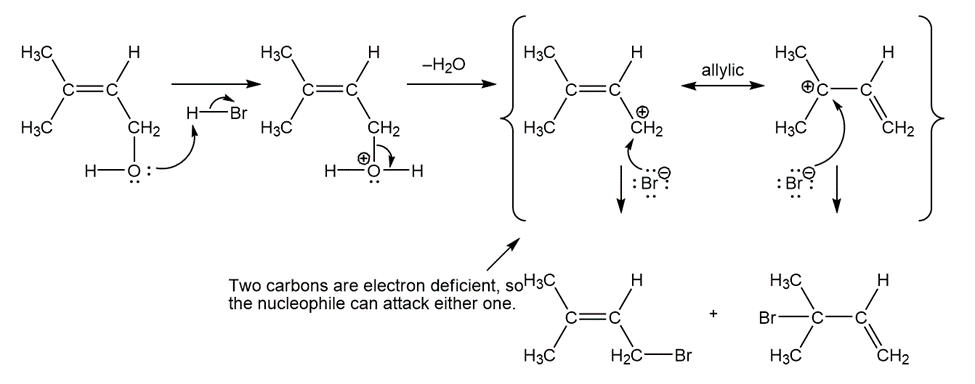
b)
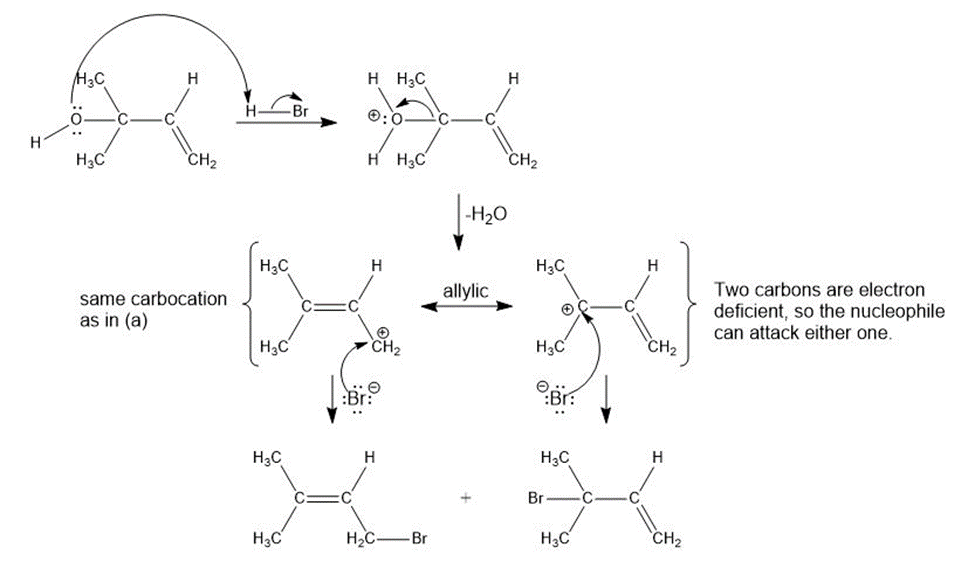
c)
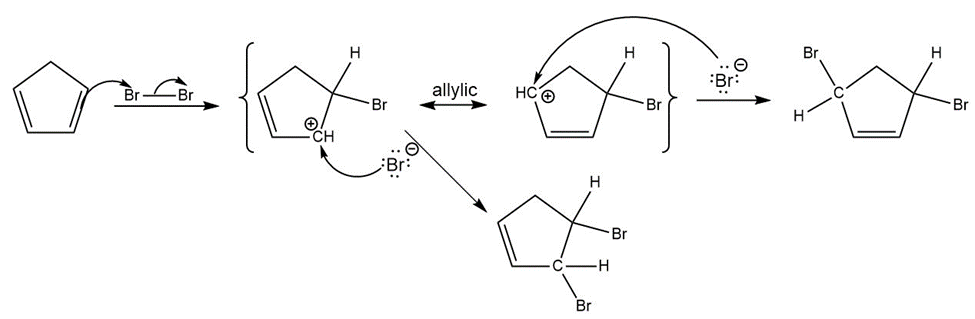
d)
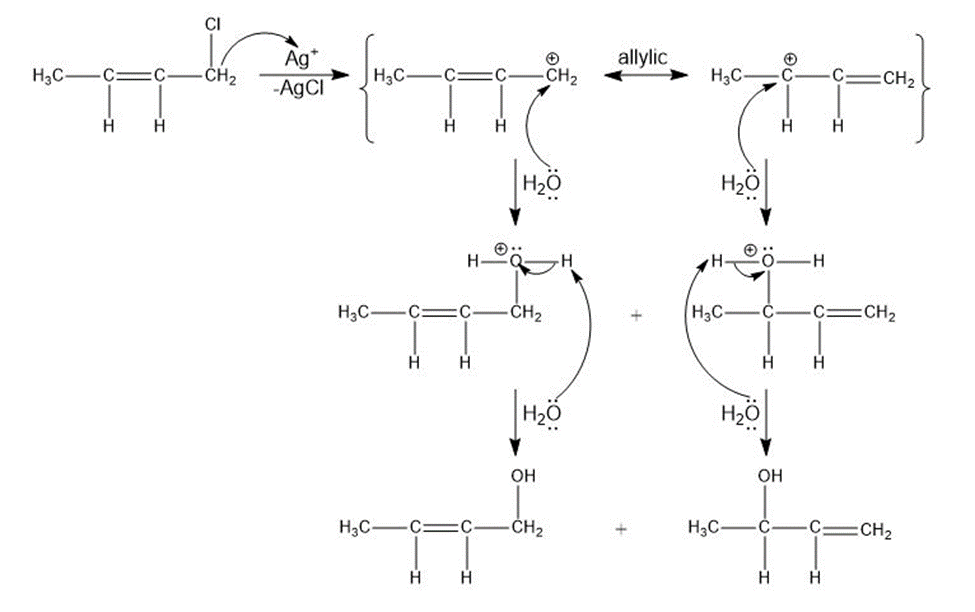
e)
Learning Materials
Features
Discover
Chapter 15: Q7P (page 761)
Propose a mechanism for each reaction, showing explicitly how the observed mixtures of products are formed.
a)
b)c)d)
e)
a)

b)

c)

d)

e)

All the tools & learning materials you need for study success - in one app.
Get started for free
What dienes and dienophiles would react to give the following Diels-Alder products?
a)
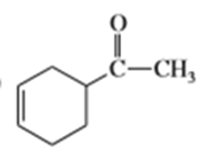
b)
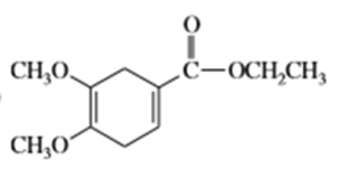
c)
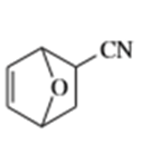
d)
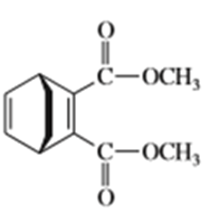
e)
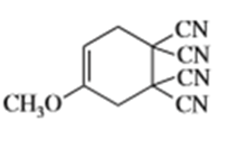
f)
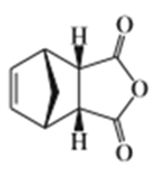
Addition of 1-bromobut-2-ene to magnesium metal in dry ether results in formation of a Grignard reagent. Addition of water to this Grignard reagent gives a mixture of but-1-ene and but-2-ene (cis and trans). When the Grignard reagent is made using 3-bromobut-1-ene, addition of water produces exactly the same mixture of products in the same ratios. Explain this curious result.
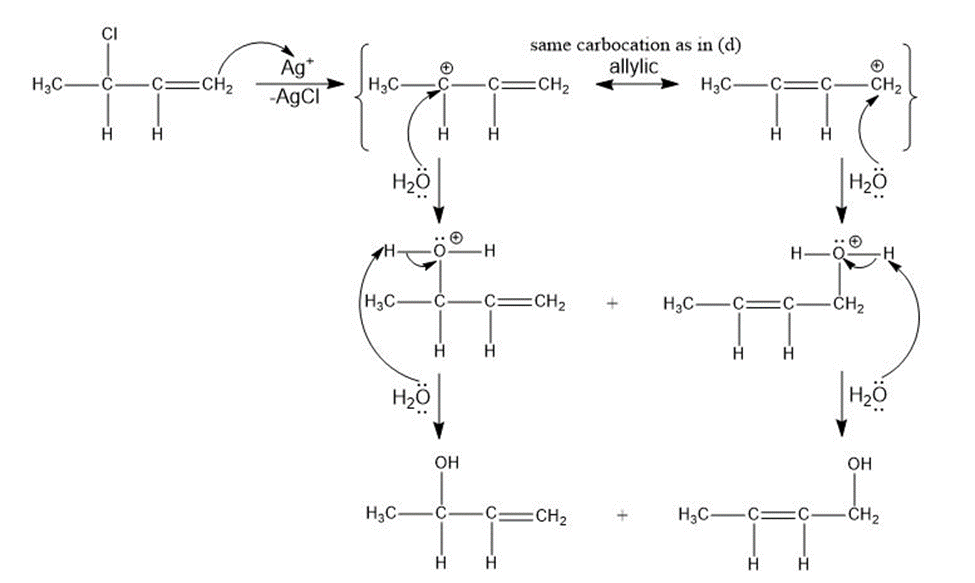
Treatment of an alkyl halide with AgNO3 in alcohol often promotes ionization.
Ag+ + R-Cl → AgCl + R+
When 4-chloro-2-methylhex-2-ene reacts with AgNO3 in ethanol, two isomeric ethers are formed. Suggest structures, and propose a mechanism for their formation.
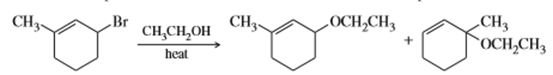
Question: When 3-bromo-1-methylcyclohexene undergoes solvolysis in hot ethanol, two products are formed. Propose a mechanism that accounts for both of these products.
Question: Predict the product(s) of light-initiated reaction with NBS in CCl4 for the following starting materials.
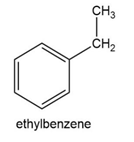
What do you think about this solution?
We value your feedback to improve our textbook solutions.