Chapter 15: 15-19P (page 782)
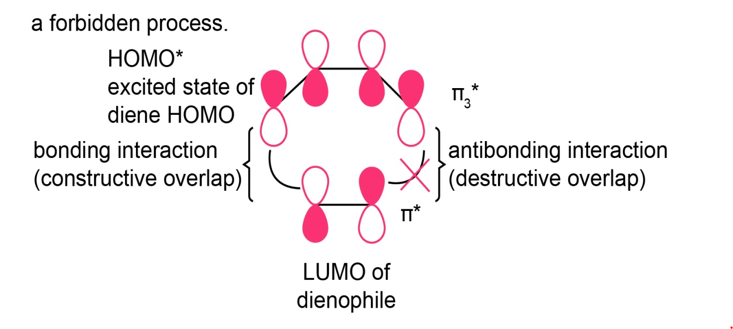
Show that the [4+2] Diels-Alder reaction is photochemically forbidden.
Short Answer
In the Diels-Alder cycloaddition, the LUMO of the dienophile and the excited state of the HOMO of the diene (HOMO*) generate one bonding and one antibonding interaction. Hence, it is a photochemically forbidden process.