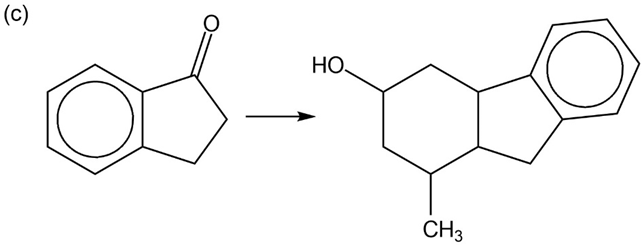
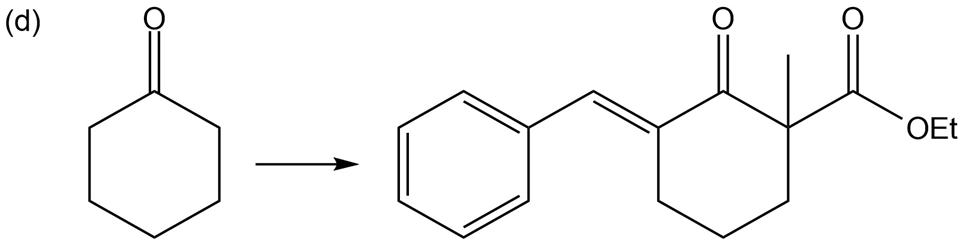
Chapter 22: Q85P (page 1207)
Show reaction sequences that explain these transformations:
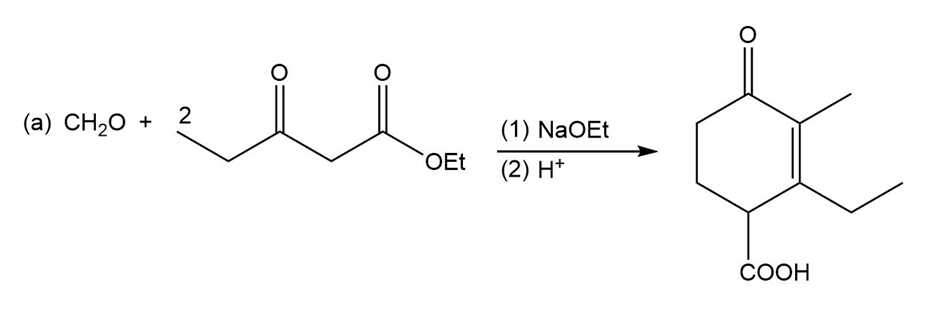
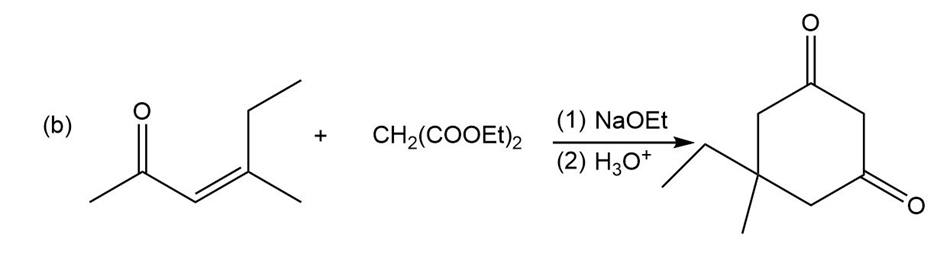
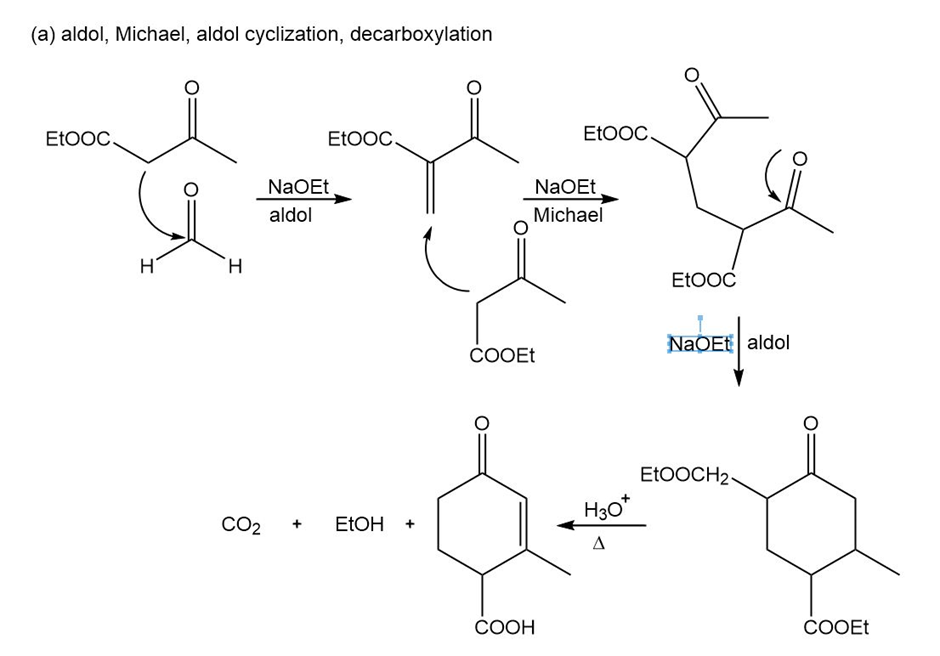
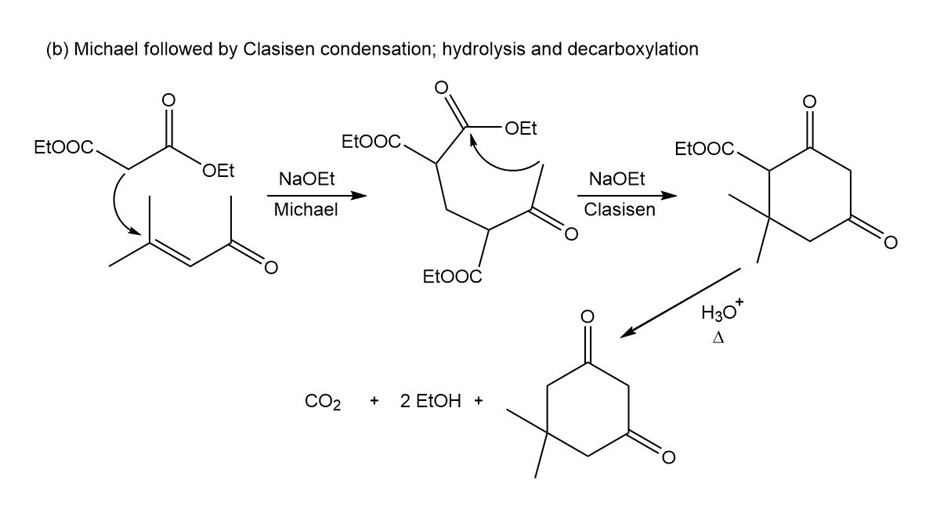
Short Answer
Learning Materials
Features
Discover
Chapter 22: Q85P (page 1207)
Show reaction sequences that explain these transformations:

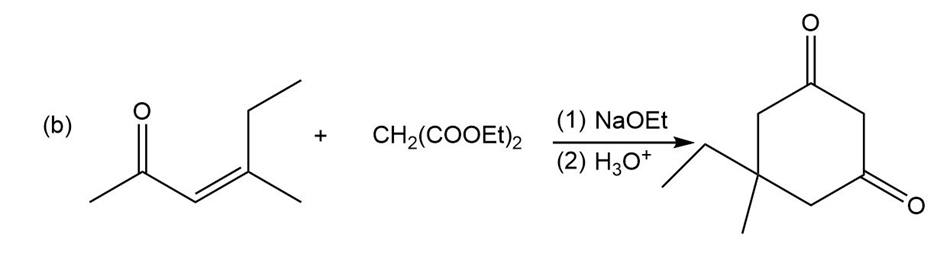


All the tools & learning materials you need for study success - in one app.
Get started for free
Propose a mechanism for the acid-catalyzed reaction of cyclohexanone with pyrrolidine.
When propionaldehyde is warmed with sodium hydroxide, one of the products is 2-methylpent-2-enal. Propose a mechanism for this reaction.
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
d.
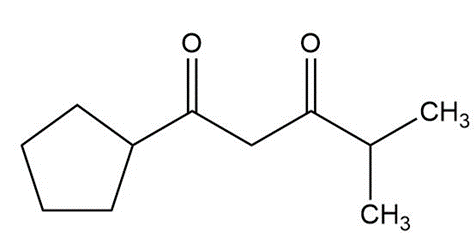
Some (but not all) of the following keto esters can be formed by Dieckmann condensations. Determine which ones are possible, and draw the starting diesters.
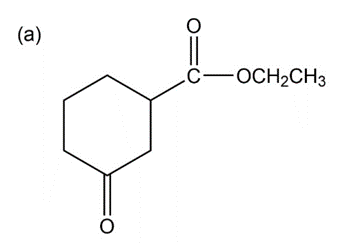
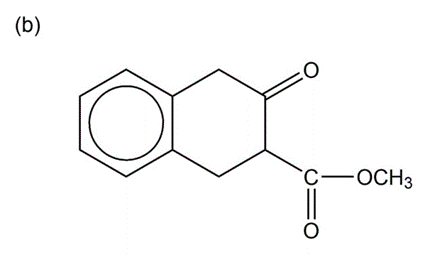
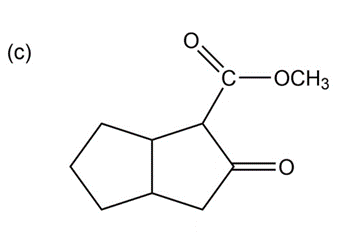
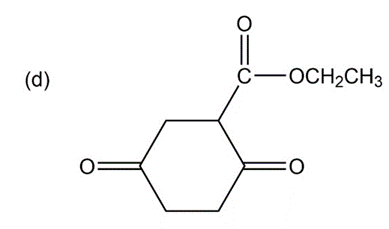
Show how you would accomplish the following multistep conversions. You may use any additional reagents you need.
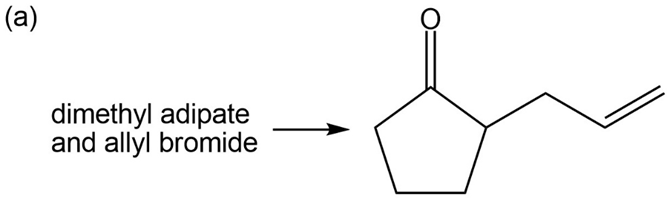
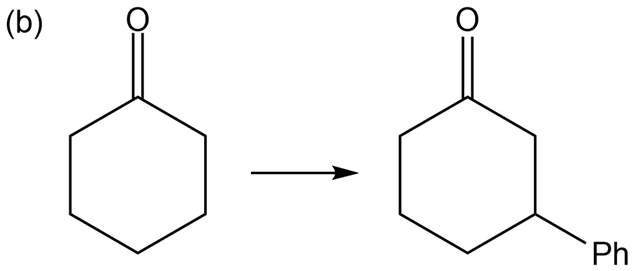
What do you think about this solution?
We value your feedback to improve our textbook solutions.