Chapter 22: Q72P-a (page 1205)
Show how you would use the malonic ester synthesis to make the following compounds
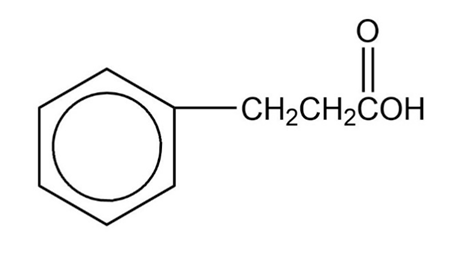
Short Answer
Learning Materials
Features
Discover
Chapter 22: Q72P-a (page 1205)
Show how you would use the malonic ester synthesis to make the following compounds


All the tools & learning materials you need for study success - in one app.
Get started for free
The following compound results from base-catalyzed aldol cyclization of a 2-substituted cyclohexanone.
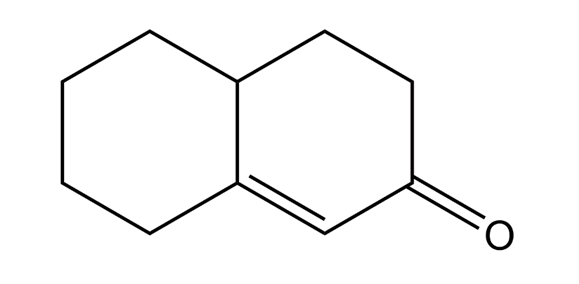
Show how octane-2,7-dione might cyclize to a cycloheptenone. Explain why ring closure to the cycloheptenone is not favored.
Show how the following ketones might be synthesized by using the acetoacetic ester synthesis.
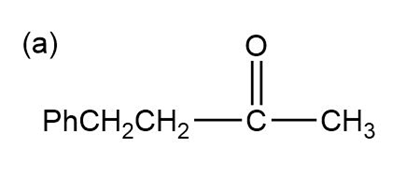
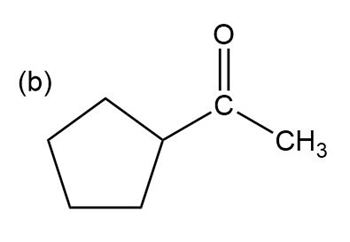
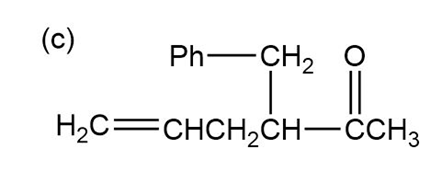
Phenylacetone can form two different enols.
Propose a mechanism for the conjugate addition of a nucleophile (Nuc:-) to acrylonitrile (H2C=CHCN) and to nitroethylene. Use resonance forms to show how the cyano and nitro groups activate the double bond toward conjugate addition.
What do you think about this solution?
We value your feedback to improve our textbook solutions.