Chapter 22: Q.68P-b (page 1204)
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
b.
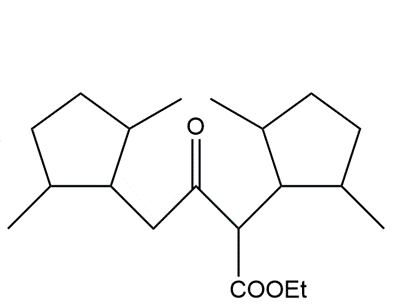
Short Answer
Answer
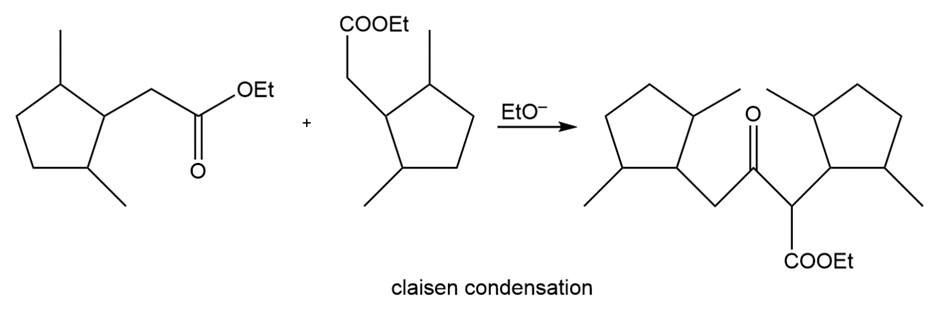
Learning Materials
Features
Discover
Chapter 22: Q.68P-b (page 1204)
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
b.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
f.
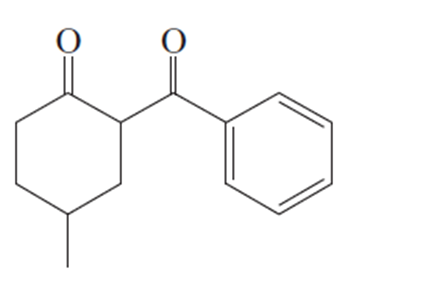
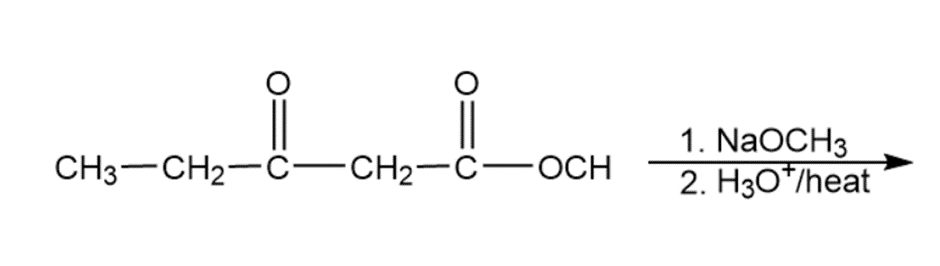
Question: Predict the products of the following reactions
h.
When cyclodecane-1,6-dione is treated with sodium carbonate, the product gives a UV spectrum similar to that of 1-acetyl-2-methylcyclopentene. Propose a structure for the product, and give a mechanism for its formation.
The Knoevenagel condensation is a special case of the aldol condensation in which an active methylene compound reacts with an aldehyde or ketone, in the presence of a secondary amine as a basic catalyst, to produce a new C=C. Show the starting materials that made each of these by a Knoevenagel condensation.
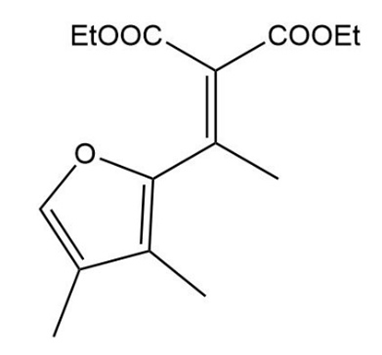
Show how you would use the acetoacetic ester synthesis to make the following compounds
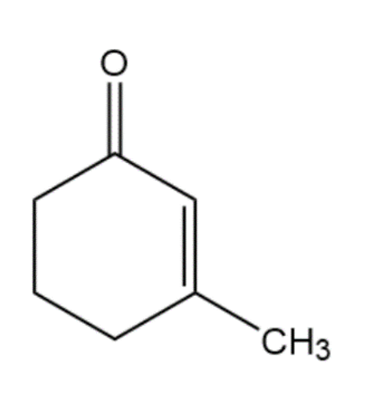
What do you think about this solution?
We value your feedback to improve our textbook solutions.