Chapter 22: Q12P (page 1161)
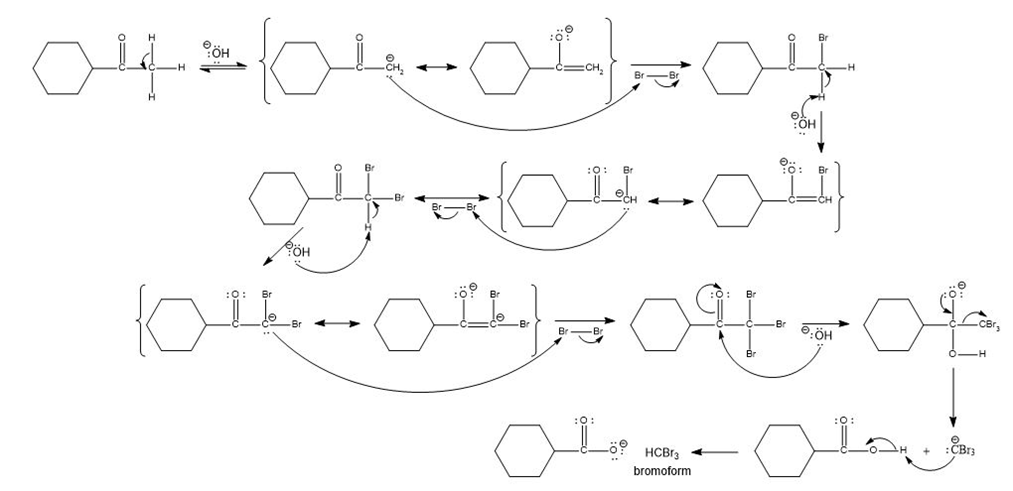
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.
Short Answer
Learning Materials
Features
Discover
Chapter 22: Q12P (page 1161)
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.

All the tools & learning materials you need for study success - in one app.
Get started for free
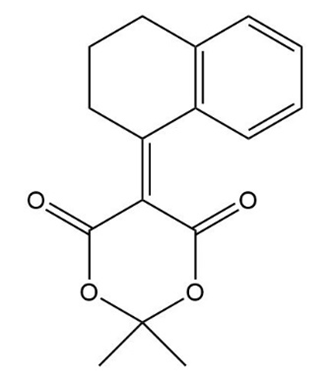
The Knoevenagel condensation is a special case of the aldol condensation in which an active methylene compound reacts with an aldehyde or ketone, in the presence of a secondary amine as a basic catalyst, to produce a new C=C. Show the starting materials that made each of these by a Knoevenagel condensation.
Predict the major products of the following reactions.
a)
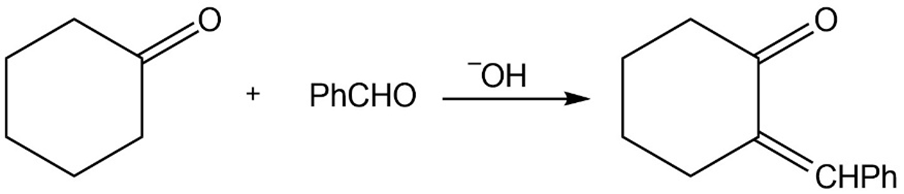
Propose mechanisms for the following reactions.
Without looking back, propose a mechanism for the hydrolysis of this iminium salt to the alkylated ketone. The first step is attack by water, followed by loss of a proton to give a carbinolamine. Protonation on nitrogen allows pyrrolidine to leave, giving the protonated ketone.
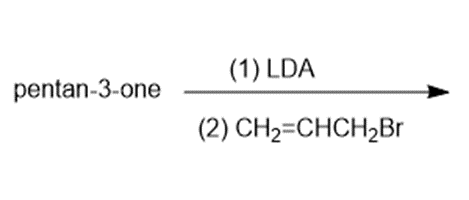
Question:Show how you would use an aldol, Claisen, or another type of condensation to make each compound
f.
What do you think about this solution?
We value your feedback to improve our textbook solutions.