Chapter 20: Q9P (page 1038)
Show how you would use appropriate acyl chlorides and amines to synthesize the following amides.
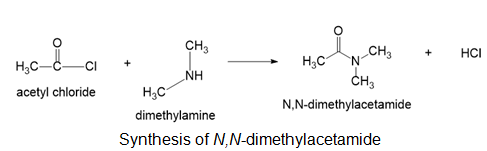
(a)N,N-dimethylacetamide
(b)acetanilide (PhNHCOCH3)
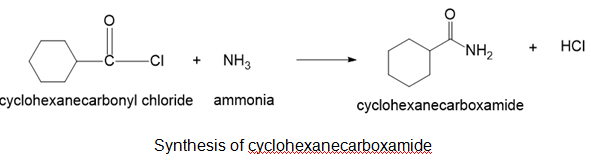
(c)cyclohexanecarboxamide
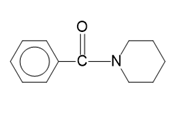
(d)
Short Answer
(a)
(b) 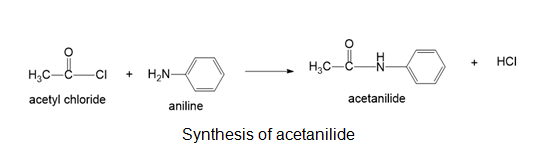
(c)
(d)
Learning Materials
Features
Discover
Chapter 20: Q9P (page 1038)
Show how you would use appropriate acyl chlorides and amines to synthesize the following amides.
(a)N,N-dimethylacetamide
(b)acetanilide (PhNHCOCH3)
(c)cyclohexanecarboxamide
(d)

(a)

(b) 
(c)

(d)

All the tools & learning materials you need for study success - in one app.
Get started for free
Glutathione (GSH) is a tripeptide that serves as a mild reducing agent to detoxify peroxides and maintain the cysteine residues of hemoglobin and other red blood cell proteins in the reduced state. Complete hydrolysis of glutathione gives Gly, Glu, and Cys. Treatment of glutathione with carboxypeptidase gives glycine as the first free amino acid released. Treatment of glutathione with 2,4-dinitrofluorobenzene (Sanger reagent, Problem 24-21, page 1282), followed by complete hydrolysis, gives the 2,4-dinitrophenyl derivative of glutamic acid. Treatment of glutathione with phenyl isothiocyanate does not give a recognizable phenylthiohydantoin, however.
(a) Propose a structure for glutathione consistent with this information. Why would glutathione fail to give a normal product from Edman degradation, even though it gives a normal product from the Sanger reagent followed by hydrolysis?
(b) Oxidation of glutathione forms glutathione disulfide(GSSG). Propose a structure for glutathione disulfide, and write a balanced equation for the reaction of glutathione with hydrogen peroxide.
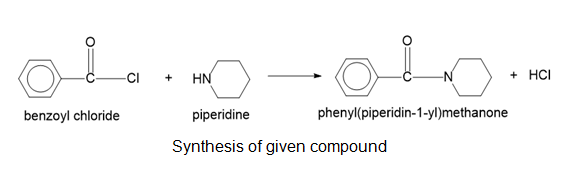
Show how you would synthesize the following compounds from the appropriate carboxylic acids or acid derivatives.
(a)
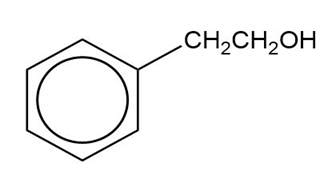
(b)
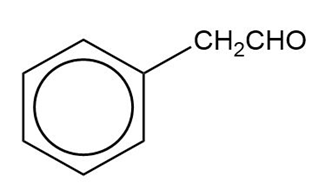
(c)
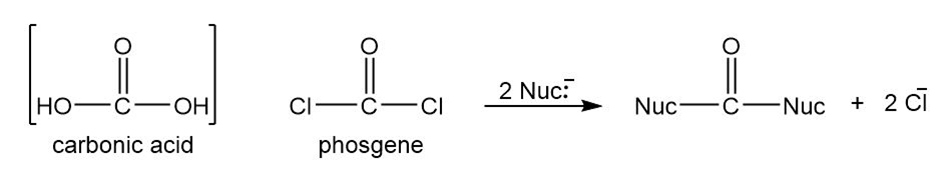
Phosgene is the acid chloride of carbonic acid. Although phosgene was used as a war gas in World War I, it is now used as a reagent for the synthesis of many useful products. Phosgene reacts like other acid chlorides, but it can react twice.
(a) Predict the products formed when phosgene reacts with excess propan-1-ol.
(b) Predict the products formed when phosgene reacts with 1 equivalent of ethanol, followed by 1 equivalent of aniline.
(c) Iso-butyloxycarbonyl chloride is an important reagent for the synthesis of peptides and proteins. Show how you would use phosgene to synthesize iso-butyloxycarbonyl chloride.
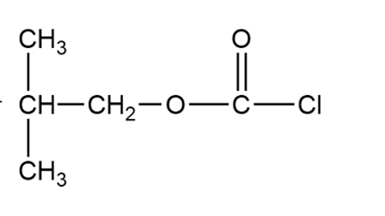
Isobutyloxycarbonyl chloride
Tributylin hydride (Bu3SnH)is used synthetically to reduce alkyl halides, replacing a halogen atom with hydrogen. Free-radical initiators promote this reaction, and free-radical inhibitors are known to slow or stop it. You job is to develop a mechanism, using the following reaction as an example.
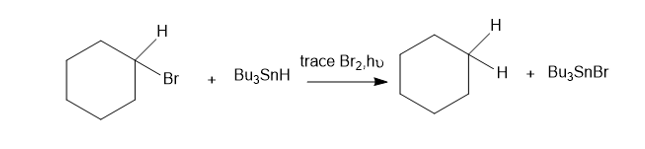
The following bond-dissociation enthalpies may be helpful:
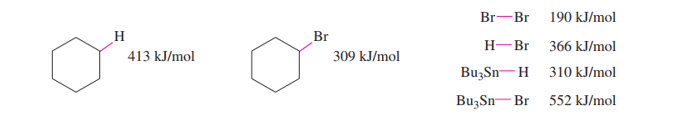
(a) Propose initiation and propagation steps to account for this reaction.
(b) Calculate values of Δ�� for your proposed steps to show that they are energetically feasible. (Hint: A trace of Br2 and light suggests it’s there only as an initiator, to create Brradicals. Then decide which atom can be extracted most favorably from the starting materials by the Brradical. That should complete the initiation. Finally, decide what energetically favored propagation steps will accomplish the reaction).
In each case, show how you would synthesize the chloride, bromide, and iodide from the corresponding alcohol.
(a) 1-halopropane (halo = chloro, bromo, iodo)
(b)1-halo-2-methylcyclopentane
(c)1-halo-1,3-dimethylcyclohexane
(d)2-halo-1,3-dimethylcyclohexane
What do you think about this solution?
We value your feedback to improve our textbook solutions.