Chapter 20: Q58P (page 1038)
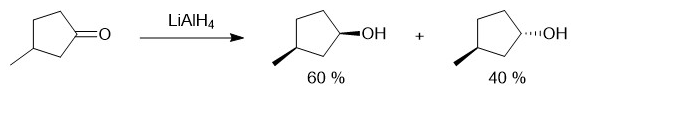
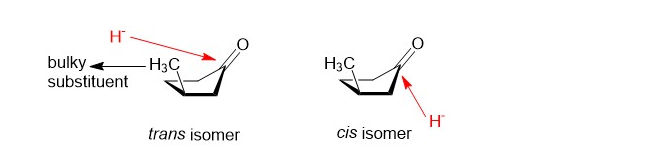
When reduces , the product mixture contains and . Use your models, and make three-dimensional drawings to explain this preference for the cis isomer
Short Answer
Learning Materials
Features
Discover
Chapter 20: Q58P (page 1038)
When reduces , the product mixture contains and . Use your models, and make three-dimensional drawings to explain this preference for the cis isomer


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons.
Show how you would synthesize the following compounds from the appropriate carboxylic acids or acid derivatives.
a)
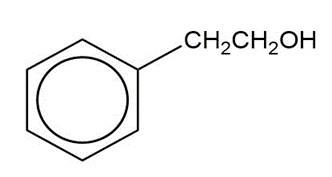
b)
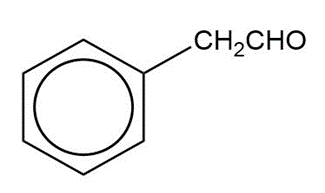
c)
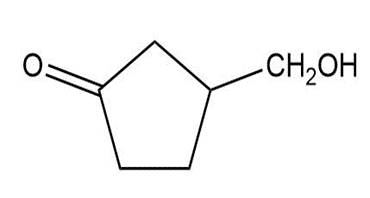
Question: Arrange each group of compounds in order of increasing basicity.
The following NMR spectra correspond to the compound of formula (A) C9H10O2, (B) C4H6O2, C6H10O2 . Propose structure, and show how it is consistent with the observed absorptions.
When pure (S)-lactic acid is esterified by racemic butan-2-ol, the product is 2-butyl lactate, with the following structure:
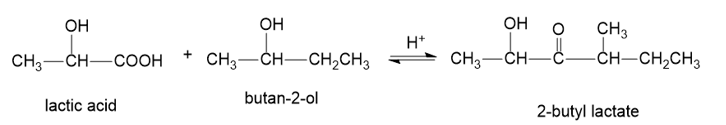
(a)Draw three-dimensional structures of the two stereoisomers formed, specifying the configuration at each asymmetric carbon atom. (Using your models may be helpful.)
(b)Determine the relationship between the two stereoisomers you have drawn.
What do you think about this solution?
We value your feedback to improve our textbook solutions.