Chapter 20: Q.47P (page 1078)
Substituent effects: resonance and induction.
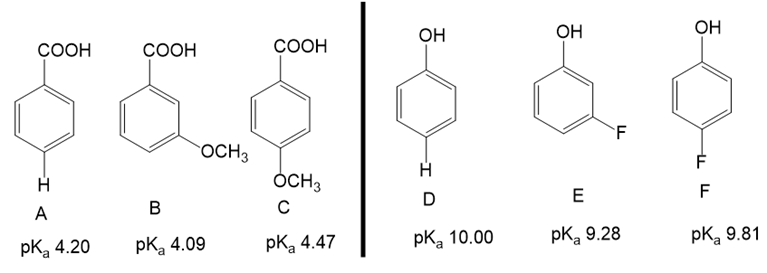
(a)When the methoxy group is in the meta position inB, is the acid stronger or weaker than inA? Is the methoxy group in the meta position electron-donating or withdrawing?
(b)When the methoxy group is in the para position inC, is the acid stronger or weaker than inA? Is the methoxy group in the para position electron-donating or withdrawing?
(c)How can this apparent contradiction be explained? Which effect is stronger for methoxy?
(d)When the fluoro group is in the meta position inE, is the acid stronger or weaker than inD? Is the fluoro group in the meta position electron-donating or withdrawing?
(e)When the fluoro group is in the para position inF, is the acid stronger or weaker than inD? Is the fluoro group in the para position electron-donating or withdrawing?
(f)Compare the results of the fluoro group with those of the above methoxy group. What must be different about the relative strength of the resonance and inductive effects for fluoro compared with methoxy?
Short Answer
(a) The methoxy group present in the meta position act as an electron-withdrawing group. So B is a stronger acid than A.
(b) The methoxy group present in the para position is electron-donating so C is a weaker acid than A.
(c) The meta substituent exerts inductive effect so it is electron-withdrawing and para position exerts resonance effect, so it is electron-donating. Thus, the resonance is stronger than induction.
(d) The fluoro group in meta position is electron-withdrawing so e is a stronger acid than D.
(e) The fluoro group is present in a para position slightly withdrawing so F is a stronger acid than D.
(f) The meta position exerts an inductive effect while the para position has a resonance effect. Thus, the resonance effect results in electron-donating but for the fluoro group, the inductive effect becomes stronger than the resonance effect.