Chapter 20: Q29P (page 1038)
Rank the following carbocations in decreasing order of stability. Classify each as primary, secondary, or tertiary.
(a)
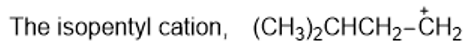
(b)
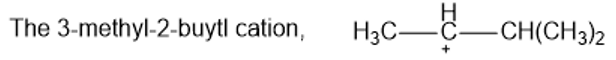
(c)
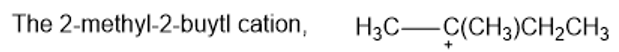
(d)
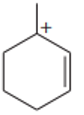
Short Answer
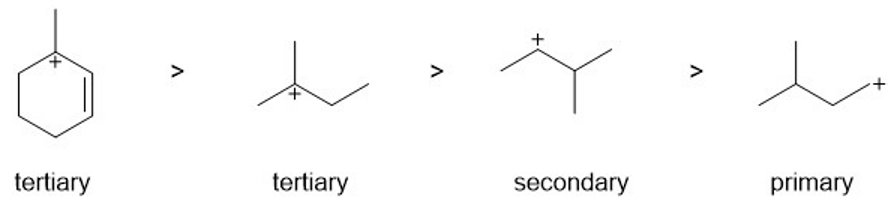
carbocations in order from most stable to least stable
Learning Materials
Features
Discover
Chapter 20: Q29P (page 1038)
Rank the following carbocations in decreasing order of stability. Classify each as primary, secondary, or tertiary.
(a)
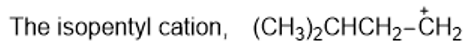
(b)
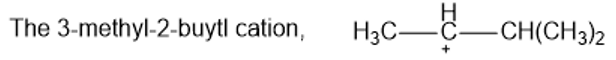
(c)
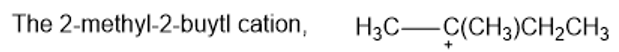
(d)


carbocations in order from most stable to least stable
All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would use extractions with a separatory funnel to separate a mixture of the following compounds: benzoic acid, phenol, benzyl alcohol, aniline.
Given the structure of ascorbic acid (vitamin C):
(a) Is ascorbic acid a carboxylic acid?
(b) Compare the acid strength of ascorbic acid (pKa=4.71) with acetic acid.
(c) Predict which proton in ascorbic acid is the most acidic.
(d) Draw the form of ascorbic acid that is present in the body (aqueous solution, pH = 7.4).
The IR, NMR, and mass spectra are provided for an organic compound.
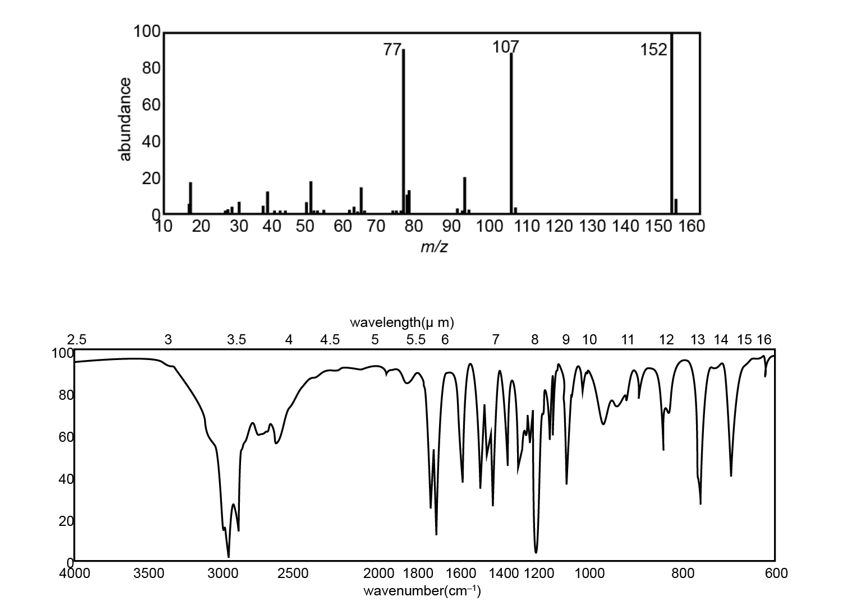
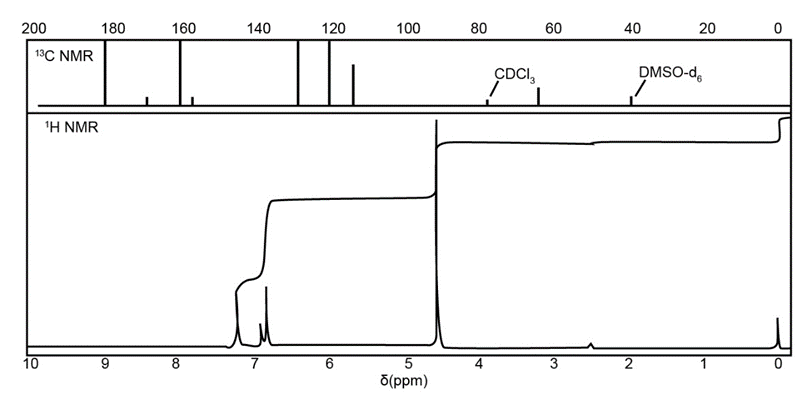
Question: Convert the following infrared wavenumbers into wavelengths.
(a) 1600 cm-1, typical for an aromatic
(b) 3000 cm-1 , typical for a saturated C-H bond
(c) 1715 cm-1 , typical for a ketone carbonyl
(d) 1750cm-1 , typical for an ester carbonyl
(e)2200cm-1 , typical for a nitrile
(f) 3300cm-1 , typical for an alcohol O-H.
(a) Propose a mechanism for the following reaction: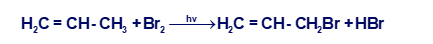
(b) Use the bond-dissociation enthalpies given in Table 4-2 (page 203) to calculate the value of ∆H° for each step shown in your mechanism. (The BDE for is about 280 kJ/mol, or 67 kcal/mol.) Calculate the overall value of ∆H° for the reaction. Are these values consistent with a rapid free-radical chain reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.