Chapter 21: Q14P (page 1107)
Propose a mechanism for the following ring-opening transesterification. Use the mechanism in Problem 21-13 as a model.
Short Answer
Mechanism of the reaction:
Learning Materials
Features
Discover
Chapter 21: Q14P (page 1107)
Propose a mechanism for the following ring-opening transesterification. Use the mechanism in Problem 21-13 as a model.
Mechanism of the reaction:
All the tools & learning materials you need for study success - in one app.
Get started for free
Name the following carboxylic acid derivatives, giving both a common name and an IUPAC name where possible.
a.
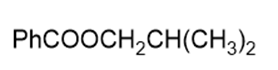
b.
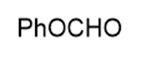
c.
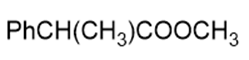
d.
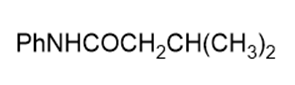
e.

f.
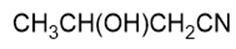
g.
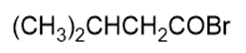
h.

i.
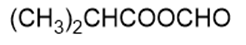
j.
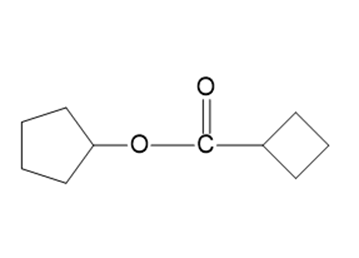
k.
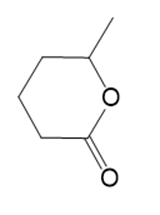
l.
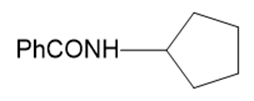
m.
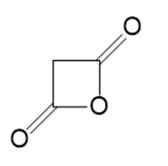
n.
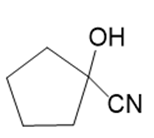
o.
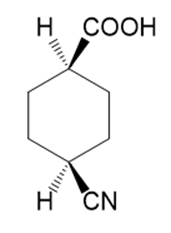
p.
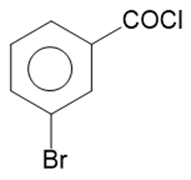
q.
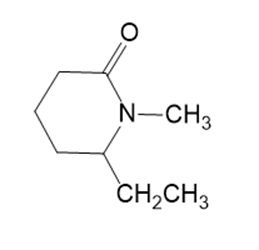
r.
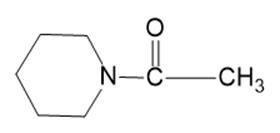
Show how you would convert the following starting materials to the indicated nitriles:
Amides, ketones, carboxylic acids, and aldehydes all give strong carbonyl stretching absorptions in the IR spectrum. How can you use other peaks in their IR spectra to distinguish among these four common functional groups?
Show how you would accomplish the following synthetic transformations. You may use any necessary reagents.
Show how you would synthesize each compound, starting with an ester containing nomore than eight carbon atoms. Any other necessary reagents may be used.
a. PH3C-OH
What do you think about this solution?
We value your feedback to improve our textbook solutions.